-
Our Products
- Aroma Chemicals
- Citronellyl Tiglate
- Cis-3 Hexenyl Butyarate
- Cis-3-Hexenyl Methyl Carbonate
- Di Methyl Octanyl Acetate
- Cyprene Ex Cypriol
- Citronellyl Propionate
- Cis-3-Hexenyl Acetate
- Citronellyl Butyrate
- Cis-3 Hexenyl Caproate
- Cis-3-Hexenyl Benzoate
- Dimethyl Benzyl Carbinol
- Citronellyl Isobutyrate
- Cinnamic Aldehyde
- Dimethyl Disulfide
- Ethyl Laurate
- Isoamyl Salicylate
- Ethyl Benzoate
- Hydratropic Aldehyde
- Ethyl Lactate
- Ethyl Maltol
- Lauryl Alcohol
- Methyl Octanoate
- Gum Arabic
- Ethyl Acrylate
- Hydratropic Alcohol
- Menthyl Acetate
- Ethyl Anthranilate
- Ethyl Formate
- Methyl 4-Methyl Benzoate
- Iso Amyl Iso Valerate
- Iso Amyl Butyrate
- Isopropyl Acetate
- Propionic Acid
- N-Butyl Anthranilate
- Sandalwood Oil
- Iso Amyl Phenyl Acetate
- Iso Butyl Phenyl Acetate
- Mint Terpenes
- Methyl Propionate
- 2-Acetyl Furan
- Pomelo Acetal
- Para Cresyl Acetate
- Methyl Propyl Ketone
- Isobutyl Isobutyrate
- Mesityl Oxide
- L-Glutamic Acid
- Methyl Chemical
- Isopropenyl Acetate
- Iso Propyl Propionate
- Styrallyl Propionate
- Methyl 2-Methyl Benzoate
- Methyl Octine Carbonate
- Isoamyl Benzoate
- Isopropyl Phenyl Butanal
- Yeast Extract
- Isobornyl Cyclohexanol
- Muguet Aldehyde
- Mushroom Alcohol
- Methyl Salicylate
- Styrallyl Alcohol
- Phenyl Ethyl Cinnamate
- Neo Menthol
- Vetiveryl Acetate
- Phenoxy Ethyl Isobutyrate
- Phenyl Ethyl Formate
- Peony Acetonitrile
- Terpinyl Acetate
- N-Caproic Acid
- Phenyl Ethyl Isobutyrate
- Di Methyl Octanol
- Phenyl Ethyl Tiglate
- Phenylethyl Bromide
- N-Propyl Acetate
- Phenyl Ethyl Phenyl Acetate
- Raspberry Ketone
- Para-Cresyl Phenyl Acetate
- Guaiyl Acetate
- Allyl Disulfide
- Phenyl Ethyl Salicylate
- Phenylacetic Acid
- Octyl Acetate
- Hydroxy Citronellol
- Triethyl Citrate
- Diallyl Disulfide
- Honey Ketone
- Isobuteric Acid
- Turmeric Oil
- Linalyl Acetate
- Tolu Balsam
- Mix Xylene
- Dementholised Oil
- Isovaleric Acid
- Methyl 2-Furoate
- Iso Butyl Salicylate
- Methoxy Melonal
- Phenethyl Acetate
- Thioacetic Acid
- Prenyl Acetate
- Verdyl Propionate
- Rose Oxide
- Tea Tree Oil
- 2 Methyl 2 Methyldithio Propanal
- 1-Butanethiol (109-79-5) (C4h10s)
- Cinnamyl Isobutyrate
- Aam Panna Flavour
- Butyric Acid
- 2-phenoxyethanol (122-99-6) (Phenyl Glycol)
- Camphor (76-22-2) (C10H16O)
- Caprylic Acid
- 4-Tert-Butyl Cyclohexanol
- Catechol (120-80-9)
- Acetoin (513-86-0)
- Amyl Acetate
- Anisole (100-66-3)
- Aceto phenone
- Allyl Isothiocyanate
- Alpha-Copaene (3856-25-5)
- Benzyl Alcohol
- Cycloh exanone
- 1,4- Dimethoxybenzene
- 1-Butanethiol (109-79-5)
- 2-Ethylhexanol (104-76-7)
- Butyl Acetate
- 2-Octanol (123-96-6)
- 3-Hydroxyacetophenone (121-71-1)
- Carvacrol (499-75-2)
- 4-Tert-Butylcyclohexyl Acetate
- 5-Decanolide (705-86-2)
- Acetoin (513-86-0)
- Amyl Acetate
- Cis-3-Hexenyl-2-Methyl Butyrate
- Acetyl Tributyl Citrate
- Alpha-Phellandrene (99-83-2)
- Benzaldehyde (100-52-7)
- Benzyl Benzoate
- Isoamyl Acetate
- Isoamyl Alcohol
- Isoborneol (124-76-5)
- Isobornyl Acetate
- Ben-zyl Cy-anide
- Isobutyraldehyde (78-84-2)
- Beta-Sitosterol (83-46-5)
- Hexyl Acetate
- Lemongrass Oil
- Cinnamon Oil
- D-Carvone (2244-16-8)
- Hydroxycitronellal (107-75-5)
- Nerol Oxide
- P-Cresol (106-44-5)
- Maltol (118-71-8)
- Melonal (106-72-9)
- Phenethyl Propionate
- Methyl 6-Methylnicotinate
- Methyl Anthranilate
- Methyl Acetoacetate
- Methyl Benzoate
- Piperonal (120-57-0)
- Methyl Eugenol
- Methyl Formate
- Methyl Heptenone
- Methyl Nic-otinate
- Methyl Phenylacetate
- Propiona ldehyde
- Alpha- Terpineol
- Benzylidene Acetone
- Beta Pinene
- Cinnamic Acid
- Coumarin (91-64-5)
- Diacetyl (0431-03-08)
- Dimethyl Sulfide
- D-Limonene (5989-27-5)
- Helional (1205-17-0)
- Heptanoic Acid
- Fruity Ketal
- 4- Methylbenzaldehyde
- Ethyl Acetoacetate
- Galaxolide (1222-05-05)
- Gamma-Decalactone (706-14-9)
- Gamma-Octalactone (104-50-7)
- Ethyl Salicylate
- Ethylene Glycol Diacetate
- Eve rnyl
- Farnesol (4602-84-0)
- Guaiacol (90-05-1)
- 6-Methylcoumarin (92-48-8)
- Anisole (100-66-3)
- Ambro cenide
- Indene (95-13-6)
- Benzyl Cy-anide
- Ethylene Glycol Monobutyl Ether
- Myristic Acid
- N-Butyraldehyde (123-72-8)
- N- Propanol
- Lavender Oil
- Lemon Oil
- Nerolidol (7212-44-4)
- Limonene (138-86-3)
- Linalool (78-70-6)
- Octanol (111-87-5)
- Lyral (31906-04-04)
- Malic Acid
- Methyl Ethyl Ke-tone
- Methyl Nonyl Ketone
- Methyl Tert-Butyl Ether
- Amber Fleur
- 1-Butanol (71-36-3)
- 2-Butanol (78-92-2)
- Butyl Butyrate
- Catechol (120-80-9)
- 5-Methyl -2-Hepten-4-One
- Benzophenone (119-61-9)
- Nootkatone (4674-50-4)
- 2- Methoxy-4-(4-Methyl-1,3-Dioxolan-2-Yl)Phenol
- Bisa bolol
- Citronella Oil
- Diethyl Malonate
- Dihydromyrcenol (18479-58-8)
- Ethyl Isobutyrate
- Ethyl Linalool
- Ethyl Nic-otinate
- Geraniol (106-24-1)
- Hedione (24851-98-7)
- Mandarin Aldehyde
- Methyl Jasmonate
- Para-Anisyl Methyl Ketone
- Resinoid Guggul
- Skatole (83-34-1)
- Salicylaldehyde (90-02-8)
- San dalore
- Thymol (89-83-8)
- Tonalide (21145-77-7)
- Vanillin (121-33-5)
- Vanillylacetone (122-48-5)
- Veratrole (91-16-7)
- Diethyl L-(+)-Tartrate
- Valencene (4630-07-03)
- 10-Undecenal (112-45-8)
- 2-Chlorobenzaldehyde (89-98-5)
- 2-Methylbutanoic Acid
- 2-Phenylethanol (60-12-8)
- 3-Methoxy-3-Methyl-1-Butanol (56539-66-3)
- Aldehyde C-10
- Aubepine (0123-11-5)
- Benzyl Mercaptan
- Beta-Citronellol (106-22-9)
- Cinnamyl Alcohol
- Diethyl Succinate
- Ethyl Butyrate
- Furfural (98-01-1)
- Heptanal (111-71-7)
- Hexanal (66-25-1)
- Indole (120-72-9)
- Ethylene Brassylate
- 2-Methoxynaphthalene (93-04-9)
- Benzyl Acetone
- Cedarwood Oil
- Amyl Salicylate
- Benzyl Acetate
- Isobutanol (78-83-1)
- 4-Methoxyphenol (150-76-5)
- delta-3-carene (13466-78-9)
- Acrolein (0107-02-08)
- 2-Hexenal (6728-26-3)
- Beta-Damascenone (23696-85-7)
- Ethyl Acetate
- Ethyl Acetate
- Eugenol (97-53-0)
- Hexanal (66-25-1)
- Indole (120-72-9)
- Iso E Super
- Isoamyl Laurate
- Methyl Cinnamate
- Tert-Amyl Alcohol
- 1,4-Dimethoxybenzene (150-78-7)
- Trans,Trans-2,4-Hexadienal
- Nonanal (124-19-6)
- Germacrene D
- Octanal (124-13-0)
- Tabanon (13215-88-8)
- Gamma-Hexanolactone (0695-06-07)
- Dl-Menthol (89-78-1)
- Ethyl Octanoate
- 2,5-Dimethylpyrazine (123-32-0)
- 2-Methyl Pyrazine
- Alpha-Hexylcinnamaldehyde (101-86-0)
- Pine Oil
- 1-Nonanol (0143-08-08)
- Cardamom Aldehyde
- Geosmin
- Ambrettolide (28645-51-4)
- Cinnamyl Isobutyrate
- 2-MethylButyraldehyde
- Cis-3-Hexenyl Lactate
- Trans-2-Hexenyl Acetate
- Geraniol 60
- Hercolyn D
- Jasmolactone (32764-98-0)
- Furfurylamine (617-89-0)
- Clove oil
- Orange oil
- Aldehyde C-18
- Ethyl Hexanoate
- Delta-Dodecalactone (713-95-1)
- Fructone (10-01-6413)
- Timber Propanol
- Alpha-Terpinyl Acetate
- Cashmeran (33704-61-9)
- Verdoracine (14374-92-6)
- Orange Aldehyde
- Methional (3268-49-3)
- Cyclotene (80-71-7)
- Citronellyl Ethoxalate
- Alpha-Humulene (6753-98-6)
- Cardamom Bark Oil
- Citronellal (106-23-0)
- L-Limonene (5989-54-8)
- Turpentine Oil
- 2,6-Dimethylpyrazine (108-50-9)
- Ambergris Ketone
- 4-Terpineol (562-74-3)
- Phenylacetaldehyde 85%
- Methyl-3-Methylthio Propionate
- Veramoss
- Indobranide
- American Peppermint Oil
- Ethyl Oleate
- Gamma-Damascone (35087-49-1)
- Menthol Crystal
- Globanone
- Sandranol
- Camphor Oil
- Butyl Formate
- Menthol Oil
- Trimethyl amine 25% aa aqueous
- Isoamyl Formate
- Isovaleraldehyde (590-86-3)
- 1-Hexanol (111-27-3)
- 1-Pentanol (71-41-0)
- 2-Acetylpyrazine (22047-25-2)
- 2-Acetylpyridine (1122-62-9)
- 2-Acetylpyrrole (1072-83-9)
- 2-Heptanol (543-49-7)
- 2-Pentanol (6032-29-7)
- 2-Propanethiol (75-33-2)
- 2'-Aminoacetophenone (551-93-9)
- 3-Hydroxybenzoic Acid
- 3-Pentanone (96-22-0)
- 3,4-Dimethoxybenzaldehyde (120-14-9)
- 4-Hydroxybenzaldehyde (123-08-0)
- 4-Methoxyacetophenone (0100-06-01)
- 4-Methylacetophenone (122-00-9)
- Aldehyde C-12 Mna (110-41-8)
- Gamma-Undecalactone (104-67-6)
- Allyl Cyclohexyl Propionate
- Alpha-Amyl Cinnamaldehyde
- Alpha-Bisabolol (515-69-5)
- Alpha-Damascone (43052-87-5)
- Alpha-Farnesene (502-61-4)
- Alpha-Isomethyl Ionone
- Anisyl Acetate
- Benzyl Salicylate
- Borneol (507-70-0)
- Camphene (79-92-5)
- Citral (5392-40-5)
- Cuminaldehyde (0122-03-02)
- Cyclopentadecanone (502-72-7)
- Cyclopentanone (120-92-3)
- Dibenzyl Ether
- Dimethyl Trisulfide
- Disodium Succinate
- Ethyl Methylphenylglycidate
- Ethyl Vanillin
- Furaneol
- Gamma-Valerolactone (108-29-2)
- Geranic Acid
- Geranyl Acetate
- Geranyl Acetate
- Hexyl Salicylate
- Isobutyric Acid
- Isophorone (78-59-1)
- Jasmone (0488-10-8)
- L-Carvone (6485-40-1)
- Longifolene (475-20-7)
- M-Cresol (108-39-4)
- Methoxyphenyl Methyl Butanone
- Methyl Acetate
- Methyl Butyrate
- Methyl Isobutyl Ketone
- Methyl Laurate
- Musk Ambrette
- Musk Ketone
- Musk Xylene
- N-Valeraldehyde (110-62-3)
- O-Cresol (95-48-7)
- Octyl Palmitate
- P-Cymene (99-87-6)
- Pentanoate (109-52-4)
- Phenetole (103-73-1)
- Phenyl Ethyl Methyl Ether
- Spilanthol (25394-57-4)
- Terpinolene (586-62-9)
- Tetramethylpyrazine (1124-11-4)
- Verdyl Acetate
- Traseolide (68140-48-7)
- Phenylacetaldehyde (122-78-1)
- Eucalyptol (470-82-6)
- Myristicin (607-91-0)
- Ethyl Levulinate
- Geranyl Acetone
- Cis-3-Hexen-1-Ol (928-96-1)
- Cis-3-Hexenyl Salicylate
- Caramel Furanone
- 2-Methoxy-4-Vinylphenol (7786-61-0)
- 3'-Methoxypropiophenone (37951-49-8)
- Safranal (116-26-7)
- Ethyl Palmitate
- Ginger Oil
- Isovanillin (621-59-0)
- Furfuryl Disulfide
- Dihydrocoumarin (119-84-6)
- Beta-Damascone (23726-91-2)
- Ethyl 2-Methylbutyrate
- Alpha-Terpinene
- Allyl Hexanoate
- 4-Methoxybenzyl Alcohol
- 3-Phenyl-1-Propanol (122-97-4)
- 2-Methylanisole (578-58-5)
- Alpha-Ionone (127-41-3)
- Ethyl Propionate
- Lysmeral (80-54-6)
- Dihydro-Beta-Ionone (17283-81-7)
- Disodium-5-Guanylate (5550-12-9)
- Delta-Damascone (57378-68-4)
- 2-Acetylthiophene (88-15-3)
- 2-Heptylcyclopentanone (0137-03-01)
- Acetal (105-57-7)
- 2,4-Decadienal (25152-84-5)
- 4-Methyl-5-Thiazoleethanol (137-00-8)
- Perillaldehyde (2111-75-3)
- Styrallyl Acetate
- Sandalrome (28219-61-6)
- Trans-2-Hexen-1-Ol (928-95-0)
- Juniper Berries
- Iso-Damascone (39872-57-6)
- Isoeugenol (97-54-1)
- Isolongifolene (1135-66-6)
- Ethyl Cinnamate
- Furfuryl Mercaptan
- Civetone (542-46-1)
- Benzyl Isothiocyanate
- Anethole (104-46-1)
- Dihydromyrcene (2436-90-0)
- Benzyl Cinnamate
- Caryophyllene Oxide
- Allyl Amyl Glycolate
- 2-Isobutylthiazole (18640-74-9)
- 4-Methoxy-2-Methyl Butane Thiol
- Ethyl Phenylacetate
- Trans-Cinnamaldehyde (14371-10-9)
- 3-Methyl-2-Buten-1-Ol (556-82-1)
- Alpha-Cedrene (469-61-4)
- 3-Methylpyridine (108-99-6)
- Trans,Cis-2,6-Nonadienal (557-48-2)
- Undecavertol
- Fenchyl Acetate
- Lavandulyl Acetate
- Isolongifolene Ketone
- Jasmin Lactone
- Beta-Caryophyllene (87-44-5)
- D-Camphor (464-49-3)
- Dextro-Dihydrocarvone (7764-50-3)
- Cyclohexenone (930-68-7)
- Cyclopentanol (96-41-3)
- Disodium 5-Inosinate
- Dihydrojasmone (1128-08-01)
- Cyclohexyl ethyl acetate
- Butyl Propionate
- Amyris Oil
- Alpha-Thujone (546-80-5)
- 2,3-Dimethylpyrazine (5910-89-4)
- 2-Octanone (111-13-7)
- Synthetic Musk
- Jaggery Flavor
- Benzyl Formate
- Methoxyhexane Thiol
- Cedrat Oil
- Geranyllinalool (1113-21-9)
- Cedrat Oil
- 1-Octen-3-One (4312-99-6)
- 1-Octen-3-yl Acetate (2442-10-6)
- Isoamyl Methyl Ketone
- Aldron
- 2-Acetonaphthone 993-08-3)
- 2-Methylbutylamine (96-15-1)
- Phenyl Ethyl Allyl Ether
- Methyl 10-Undecenoate
- Vinyl Sulfurol
- Timber Touch
- Alpha-Methyl Cinnamaldehyde
- Dimethyl Anthranilate
- Tiglic Acid
- 3,5,5-Trimethyl-1-Hexanol (3452-97-9)
- Farnesal (19317-11-4)
- Methyl levulinate
- Dodecanal (112-54-9)
- Exaltolide (0106-02-05)
- Vertofix
- Orcinol Dimethyl Ether
- Bornyl Acetate
- Saffron Oil
- Delta-Octalactone (698-76-0)
- Octahydrocoumarin (4430-31-3)
- 3-Methoxy-3-Methyl Butanol
- Amyl Isovalerate
- Ethyl Isovalerate
- Methyl Cedryl Ether
- Ethyl Isoamyl Ketone
- Methyl Heptanoate
- Anthamber Top
- Exaltenone (14595-54-1)
- Glyceryl Tribenzoate
- Verdol
- 2-MethoxyBenzoic Acid
- Geranodyle
- Hydroxyambran (118562-73-5)
- Mugetanol
- Lemon Hexadiene
- Alpha-DihydroIonone (31499-72-6)
- Methyl Ionone Alpha Extra
- 4-tert-ButylCyclohexanone (98-53-3)
- Laevo-Camphor (464-48-2)
- Ethyl Caprate
- Diethyl Adipate
- Cetalox
- Trans-3-Hexenoic Acid (1577-18-0)
- 2-Isopropyl-N,2,3-Trimethylbutyramide (51115-67-4)
- 5-Methylfurfural (620-02-0)
- Theaspirane (36431-72-8)
- Aurantiol (89-43-0)
- Beta-Santalene (511-59-1)
- Isophytol (505-32-8)
- Cis-3-Hexenyl Isobutyrate
- Cis-3-Hexenyl Isobutyrate
- Veloutone
- Sandal Butenol
- 4-Methyl Guaiacol
- Lilyflore
- 2-Isopropyl-4-Methylthiazole (15679-13-7)
- Ethyl Isothiocyanate
- Hay Absolute
- 4-Methyl Thio Benzyl Cyanide
- Trans-2-Hexenoic Acid
- Industrial Chemicals
- Ammonium Carbonate
- Acetone Cyanohydrin
- Calcium Carbonate
- Chlorinated Paraffin Wax
- Calcium Chloride
- Citric Acid Monohydrate
- Ammonium Stearate
- Ammonium Polyacrylate
- Calcium Silicate
- Copper Carbonate
- Alpha Picolinic Acid
- Boron Nitride
- Calcium Oxide
- Calcium Sulfate
- Aluminium Phosphate
- Aluminium Acetate
- Alcohol Ethoxylate
- Calcium Stearate
- Cyanoacetic Acid
- Ammonium Bifluoride
- Choline Bitartrate
- Sodium Ethoxide
- Caustic Soda Flakes
- Calcium Chloride Dihydrate
- Ammonium Sulfamate
- Capric Acid
- Cobalt Sulphate
- Sodium Chloride
- Sodium Stannate
- Sodium Hydroxide
- Sodium Polyacrylate
- Sodium Metabisulfite
- Sodium Gluconate
- Sodium Propionate
- Sodium Silicate
- Melamine Cyanurate
- Zirconium Oxide
- Sodium Methoxide
- Magnesium Glycinate
- Sorbitan Trioleate
- Non Ferric Alum
- Magnesium Sulphate
- Sodium Salicylate
- Glutaric Acid
- Sodium Hydrosulfide
- Sodium Cryolite
- Glyoxylic Acid
- Magnesium Nitrate
- Lithium Carbonate
- Sodium Silicofluoride
- Zinc Hydroxide
- Magnesium Fluoride
- Sodium Thiosulphate
- Sodium Erythorbate
- Lactose Monohydrate
- Sodium Lauryl Sulfate
- Sorbitan Monooleate
- Lactic Acid
- Hydrochloride Chemical
- Methyl Ethyl Ketone Peroxide
- Mono Sodium Phosphate
- Hydrofluoric Acid
- Methyl Ethyl Ketoxime
- Hydrazine Hydrate
- Magnesium Carbonate
- Calcium Lactate
- Neopentyl Glycol
- Polyethylene Glycol
- Magnesium Oxide
- Magnesium Stearate
- Hydrogen Peroxide
- Methylene Chloride
- Phosphorus Pentoxide
- Methylamine Hydrochloride
- Guanidine Hydrochloride
- Magnesium Silicate
- Boric Acid
- Hydrochloric Acid
- Tricalcium Phosphate
- Fumaric Acid
- Zinc Stearate
- Zinc Carbonate
- Titanium Dioxide
- Triphenyl Phosphite
- Sodium Sulphate
- Silicone Emulsion
- Isopropyl Alcohol
- Boron Carbide (12069-32-8)
- Silica Gel
- Itaconic Acid
- Xanthan Gum
- Zinc Phosphate
- Sodium Formate
- Methyl Acrylate
- Copper Nitrate
- Silicon Dioxide
- Sodium Nitrate
- Nitric Acid
- Phthalic Acid
- Triphenyl Phosphate
- Hydrofluoric Acid 20%
- Hydrofluoric Acid 40%
- Aluminium Fluoride Anhydrous
- Sodium Hexafluoroaluminate
- Aluminium Fluoride Trihydrate
- Ammonium Fluoride
- Potassium Aluminium Fluoride
- Sodium Bifluoride
- Barium Carbonate
- Barium Hydroxide Anhydrous
- Barium Hydroxide Octahydrate
- Barium Hydroxide Monohydrate
- Sodium Hydrosulfide Hydrate
- Sodium Hydrosulfide Anhydrous
- Phenylhydrazine Hydrochloride
- 5-Nitroisophthalic Acid
- Acetamide (60-35-5)
- 3-Nitrobenzaldehyde (99-61-6)
- Potassium Hexafluoroaluminate
- Potassium Fluoride Anhydrous
- Ammonium Tetrafluoroborate
- Fluoroboric Acid
- Ammonium Fluorosilicate
- Hexafluorosilicic Acid
- Barium Nitrate
- Magnesium Fluoride
- Sodium fluoride
- Tin Fluoride
- Sodium Tetrafluoroborate
- Ammonium Bifluoride
- Barium Peroxide
- Phenylhydrazine (100-63-0)
- Phthalimide (85-41-6)
- Benzoguanamine (91-76-9)
- Pyridinium Chlorochromate
- Pyridine Sulfur Trioxide
- Mal-eic Hydr-azide
- 2-Bromoethylamine Hydrobromide
- 4-Phenylbutyric acid
- 3-Indoleacetic acid
- N-Methylphthalimide (550-44-7)
- 4-Dimethylaminobenzaldehyde (24057-28-1)
- Pyridinium P-Toluenesulfonate
- Potassium Bifluoride
- Potassium Tetrafluoroborate
- Barium Sulphide
- 3-Nitrophthalic Acid
- 4-Nitrophthalic Acid
- Mucic Acid
- 4-Chlorophenylhydrazine Hydrochloride
- Potassium Phthalimide
- N-2-Hydroxyethyl Phthalimide
- N-Hydroxymethyl Phthalimide
- Homophthalic acid
- Homophthalic Anhydride
- Dl-Camphorquinone (10373-78-1)
- 2-Benzylacrylic Acid
- Triphenylphosphine Oxide
- Dibenzyl Phosphate
- Cobalt Chloride Anhydrous
- Cobalt Chloride Hexahydrate
- Cobalt Nitrate (10141-05-06)
- Cobalt Sulfate Anhydrous
- Cobalt Sulphate Heptahydrate
- Zirconium Octoate (C32H64O8Zr)
- Zirconium Sulphate Anhydrous
- 2,4-Dichlorophenol (120-83-2)
- Biphenyl (92-52-4) (C12H10)
- 1,4-Naphthoquinone (130-15-4) (C10H6O2)
- Borax Pentahydrate (11130-12-4)
- 2-Cyanopyridine (100-70-9)
- 2-Mercaptopyridine N-Oxide Sodium Salt
- 2-Methyl-4-Nitroaniline (99-52-5) (C7H8N2O2)
- 2,5-Dichloroaniline (95-82-9)
- Calcium Acetate Anhydrous
- 3-Bromophenol (591-20-8)
- Calcium Carbide (75-20-7)
- Calcium Chloride Anhydrous
- Calcium Silicide (12013-56-8)
- Calcium Tungstate (7790-75-2)
- 2-Aminopyridine (504-29-0)
- 2-Ethylanthraquinone (84-51-5) (C16H12O2)
- 2-Methylimidazole (693-98-1) (C4H6N2)
- 3-Nitrobenzoic Acid (121-92-6)
- 4-Bromoaniline (106-40-1)
- 4-Chlorobenzaldehyde (104-88-1)
- 4-Chlorophenol (106-48-9) (C6H5ClO)
- 4-Hydroxybenzophenone (1137-42-4) (C13H10O2)
- 4-Nitroaniline (0100-01-06) (C6H6N2O2)
- 4-Nitrobenzaldehyde (555-16-8)
- 4-tert-Butylbenzoic acid
- Ammonium Benzoate (1863-63-4)
- Ammonium Bicarbonate
- Ammonium Bromide
- Ammonium Sulphite (10196-04-0)
- Ammonium Thiocyanate (1762-95-4)
- Acetanilide (103-84-4)
- Acetoacetanilide (0102-01-02) (C10H11NO2)
- Ammonium Thiosulphate (7783-18-8)
- Anthraquinone (84-65-1) (C14H8O2)
- Barium Chloride (10361-37-2)
- Alginic Acid
- Barium chromate (10294-40-3)
- Aluminum Acetylacetonate (13963-57-0)
- Aluminum Nitride (24304-00-5)
- Benzenesulphonic Acid
- Benzhydrol (91-01-0)
- Ammonium Molybdate (H8MoN2O4)
- Ammonium Persulphate
- Benzyltriethylammonium Chloride (56-37-1)
- Cryptohalite (16919-19-0) (Ammonium Fluorosilicate)
- Hexachloroethane (67-72-1) (C2Cl6)
- Hydroxylammonium Sulfate
- Lead Dioxide
- Lead Nitrate
- Lead Zirconium Titanate
- Nickel Acetate Anhydrous
- Nickel Chloride
- Nickel Sulfate Anhydrous
- Limestone (1317-65-3)
- Nitrilotriacetic Acid
- Lithium Acetate Dihydrate
- Lithium Chloride Anhydrous
- Lithium Fluoride Anhydrous
- Lithium Hydroxide Anhydrous
- Lithium Iodide Anhydrous
- O-Aminophenol (95-55-6)
- O-Phenylenediamine (95-54-5)
- Lithium Nitrate Anhydrous
- O-Toluic Acid
- M-Toluic Acid
- Magnesium Chloride Anhydrous
- Oxalic Acid Anhydrous
- Para Toluenesulphonic Acid Anhydrous
- Malonic Acid
- Manganese Chloride Anhydrous
- Mercuric Chloride
- Mercuric Nitrate Monohydrate
- Picramic Acid
- Picric Acid
- Potassium Permanganate
- Mica (12001-26-2)
- 3-Nitrochlorobenzene (121-73-3)
- Pumice (1332-09-08)
- Pyridine-3-Sulfonyl Chloride
- Pyridinium Dichromate
- Aluminium Nitrate
- Ammonium Bisulfite
- Chrome Alum
- Chromic Acid
- Borax Anhydrous
- Cis Bromo Benzoate
- Copper Acetate
- Copper Chloride
- Copper Naphthenate
- Copper Nitrate Trihydrate
- Cupric Chloride
- Cyanuric Chloride
- Chlorhexidine (55-56-1)
- Chloroacetic Acid
- Denatonium Benzoate
- Dimethylamine Hydrochloride
- Dimethylglyoxime (95-45-4)
- Dimethylurea
- Diphenyl Diselenide
- Dipicolinic Acid
- Dolomite (16389-88-1)
- Gallic Acid Anhydrous
- Ethylene Diamine Acid Phosphate
- Glutaric Anhydride
- Ethylene Glycol Distearate
- Ethylenediaminetetraacetic Acid
- Graphite (7782-42-5)
- Guanidine Nitrate
- Ferric Alum Dodecahydrate
- Ferric Nitrate
- Hydrazine Sulphate
- Fluorspar (7789-75-5)
- Potassium Acetate
- Potassium Aluminium Sulphate
- Potassium Bromate
- Potassium Chromate
- Potassium Ferrocyanide Anhydrous
- Potassium Fluoride Anhydrous
- P-Toluenesulfonic acid
- Sodium Amide
- Sebacic Acid
- Sodium Azide
- Sodium Bisulphate Anhydrous
- Sodium Bisulphite
- Sodium Bromide Anhydrous
- Sodium Chromate
- Sodium Tert-Pentoxide
- Sodium Hydride
- Sodium Iodide Anhydrous
- Sodium Thiosulphate Anhydrous
- Quadrol (102-60-3)
- Silver Nitrate
- Sodium Cyanate
- Sodium Tartrate
- Tetrabutylammonium Fluoride Anhydrous
- Tetrabutylammonium Fluoride Trihydrate
- Tetrachlorophthalic Anhydride
- Strontium Chloride Anhydrous
- Strontium Fluoride
- Strontium Nitrate
- Succinic Acid
- Succinic Anhydride
- Sucrose (57-50-1)
- Tolyltriazole (29385-43-1)
- Tosyl Chloride
- Trichloroacetic Acid
- Terephthalic Acid
- Vanadyl Sulfate Anhydrous
- Triphenylphosphine (603-35-0)
- Zinc Chloride Anhydrous
- 1-Tridecanol (112-70-9)
- 2-Chlorobenzoic Acid
- 2-Cyanoacetamide (107-91-5)
- 2-Naphthol (135-19-3)
- 2,2-Bis Hydroxymethy Propionic Acid
- 3,5-Dinitrobenzoic Acid
- Diphenyl Amine
- Ferrocene (102-54-5)
- Iodobenzene Diacetate
- Levulinic Acid
- Mercury Sulfide
- Octadecane (593-45-3)
- Sodium Carbonate Decahydrate
- Stannous Sulfate
- Succinimide (123-56-8)
- Tetraethylammonium Chloride
- Triethylamine Hydrochloride
- Calcium Naphthenate
- 2,6-Dichloroaniline (608-31-1)
- Sodium 2-Ethylhexanoate
- Dimethyl Aminoterephthalate
- 4-Methylbenzoic Acid
- Vanadium Trioxide
- Sodium Vinylsulfonate
- Magnesium Chloride Hexahydrate
- 3-Aminocrotononitrile (1118-61-2)
- Iodic Acid
- Strontium Chloride Hexahydrate
- Sodium Dichromate Dihydrate
- 4-Isopropylcatechol (2138-43-4)
- Tetramethylammonium Hydroxide
- Meta Nitro Para Toluidine
- Lead Acetate Trihydrate
- 4-Chloro-2-Nitroaniline
- Lithium Bromide Anhydrous
- Neodecanoic Acid
- Potassium Hydrogen Oxalate
- Sodium DiethylDithioCarbamate
- Iron Sulfate Heptahydrate
- Sodium Hydroxide Pellets AR
- 2-Amino-4-Chlorodiphenyl Ether
- Oxalic Acid Dihydrate
- Sodium Dichromate Anhydrous
- Para-Toluenesulphonic Acid Monohydrate
- 4-Aminoveratrole 6315-89-5
- Sodium Acetate
- 1,3-Benzenedisulfonic Acid
- 3-Hydroxy-N-Phenyl-2-Naphthamide (92-77-3)
- Leucoquinizarin (476-60-8)
- Magnesium Ingots
- Ferric Alum
- Alkyl Benzene Sulfonate
- Palmitoyl Chloride (112-67-4)
- Cobalt Naphthenate (61789-51-3)
- 1,2-Diaminopropane (78-90-0) (C3H10N2)
- 1,4-Butynediol (110-65-6) (C4H6O2)
- Bis(2-Ethylhexyl) Hydrogen Phosphate
- 2-Chloroethanol (0107-07-03) (C2H5ClO)
- 2-Chloropropionyl Chloride
- 2-Ethylaniline (578-54-1) (C8H11N)
- Bromoform (75-25-2) (CHBr3)
- 2-Ethylhexyl Nitrate (27247-96-7)
- 2-Mercaptoethanol (60-24-2) (C2H6OS)
- 2-Methoxyethanol (109-86-4) (C3H8O2)
- 2,4,6-Trimethylpyridine (108-75-8) (C8H11N)
- 3-2-Aminoethylamino Propyl Trimethoxysilane
- 3,5,5-Trimethylhexanoyl Chloride
- 4-Bromotoluene (106-38-7)
- 4-Heptanone (123-19-3) (C7H14O)
- 4-Methylmorpholine N-Oxide (7529-22-8)
- 4-Nitrotoluene-2-Sulfonic Acid (0121-03-09)
- Carbon Disulfide (75-15-0)
- Ammonium Hydroxide
- Acetic Acid (64-19-7)
- Acetonitrile (75-05-8) (C2H3N)
- EDTA Tetrasodium Salt Anhydrous
- Ethyltriphenylphosphonium Iodide
- Hydrocarbon Resin
- Butyltriphenylphosphonium Bromide
- Potassium Silicofluoride
- Manganese Monoxide
- 1,3-Dihydro-2h-Benzimidazole-2-Thione (583-39-1)
- Sodium Isopropoxide
- Silicotungstic Acid
- Ammonium Heptamolybdate
- Acetyl Chloride (75-36-5)
- Acetylacetone (123-54-6) (C5H8O2)
- Allyl Alcohol (107-18-6)
- Allyl Chloride (0107-05-01)
- Benzotrichloride (98-07-7) (C7H5Cl3)
- Benzoyl Chloride
- Benzyl Chloride (100-44-7)
- Isobutyl Bromide
- Isobutyric Anhydride
- Isopropylamine (75-31-0)
- Gamma-Butyrolactone (96-48-0) (C4H6O2)
- Hexamethyldisilazane (999-97-3) (C6H19NSi2)
- Monoethylene Glycol
- Morpholine (110-91-8) (C4H9NO)
- N-Butyl Chloride (109-69-3)
- N-Butylamine (109-73-9) (C4H11N)
- N-Butyllithium (109-72-8)
- N-Hexane (110-54-3)
- Lauroyl Chloride
- N-Pentane (109-66-0)
- Nitrobenzene (98-95-3)
- Nonylphenol (84852-15-3)
- Octylamine (111-86-4)
- M-Toluidine (108-44-1)
- Oleum (8014-95-7)
- Isobutylamine (78-81-9)
- Hexafluorophosphoric Acid (16940-81-1)
- Para-Chlorobenzoyl Chloride
- Petroleum Ether
- Mesitylene (108-67-8)
- Methanesulfonic Acid
- Phosphorus Oxychloride
- Methyl 2-Chloropropionate
- Phosphorus Trichloride
- Methyl Chloroformate
- Methyl Iodide
- Methyl Isonicotinate
- Methyl Isopropyl Ketone
- Methyl Ricinoleate
- Pyridine (110-86-1)
- 1 2-Dichloroethane (0107-06-02)
- 18-Crown-6 (17455-13-9)
- Aminoethyl Ethanolamine
- Chloromethyl Methyl Ether
- Chlorosulfonic Acid
- Boron Trifluoride Etherate
- Cis-2-Butene-1,4-Diol (6117-80-2)
- Calcium Petroleum Sulfonate
- Cresylic Acid
- Cumene Hydroperoxide
- Cyclohexane (110-82-7)
- Chloroacetyl Chloride
- Dibutylamine (111-92-2)
- 5-Chlorovaleroyl Chloride
- Acetyl Bromide (506-96-7)
- Monoethylamine (75-04-7) (C2H5NH2)
- N-N-N-N-Tetrakis 2-Hydroxypropyl Ethylenediamine
- Naphthenic Acid
- Oleylamine (112-90-3)
- Methyl Chloroacetate
- Propylamine (0107-10-8)
- Alkyl Phenol Ethoxylate
- Chlorine Dioxide
- Methyl Vinyl Ketone
- Dichloromethane (75-09-2)
- Diethanolamine (111-42-2)
- Diethyl Carbonate
- Diiodomethane (75-11-6)
- Diacetone Alcohol
- Diisopropanolamine (110-97-4)
- Diisopropylamine (108-18-9)
- Dimethyl Amine
- Dibromomethane (74-95-3)
- Dimethyl Oxalate
- Dimethyl Phthalate
- Dimethylformamide (68-12-2)
- Epichlorohydrin (106-89-8)
- Diphenyl Oxide
- Dodecane (112-40-3)
- Dodecyl Amine
- Ethyl Bromoacetate
- Ethyl Chloroacetate
- Ethyl vinyl ether
- Ethylcyclohexane (1678-91-7)
- Diethyl Dipropylmalonate
- Diethylaminoethanol (100-37-8)
- Diacetone Alcohol
- Fluoroboric Acid
- Formic Acid
- Polyol
- Polysorbate 80
- Polyphosphoric Acid
- Octyltriethoxysilane (2943-75-1)
- Pyrrolidine (123-75-1)
- Pine Tar
- Sodium Isopropylxanthate
- Tetraethylene Pentamine
- Stearoyl Chloride
- Thionyl Chloride
- Titanium Tetrachloride
- Sulfolane (126-33-0)
- Sulphuryl Chloride
- Tributyl Phosphate
- Tert-Butyl Alcohol
- Tert-Butyl Hydroperoxide
- Tert-Butylamine (75-64-9)
- Tert-Butylbenzene (98-06-6)
- Triethyl Phosphite
- Triethylene Glycol
- Triethylene Glycol Monobutyl Ether
- Triethylene Glycol Monomethyl Ether
- Triethylsilane (617-86-7)
- Triisopropanolamine (122-20-3)
- Trimethyl Borate
- Trixylenyl Phosphate
- 1-Methylimidazole (616-47-7)
- 1,2-Dichloropropane (78-87-5)
- 1,8-Diazabicyclo-5.4.0-7-Undecene (6674-22-2)
- 2-(2-Aminoethoxy)Ethanol (0929-06-06)
- 2-Amino-2-Methyl-1-Propanol (124-68-5)
- 2-Ethylhexylamine (104-75-6)
- 2-Fluoroaniline (348-54-9)
- 2-Methyltetrahydrofuran (96-47-9)
- 3-Mercaptopropionic Acid
- 3-Methylbenzyl Chloride
- Chlorotrimethylsilane (75-77-4)
- Cyclohexyl Isocyanate
- Dibutyl Ether
- Diethylene Glycol Monobutyl Ether
- Diisobutyl Ketone
- Dowfax 2A1
- Ethylene Carbonate
- Hydroiodic Acid
- Hypophosphorous Acid
- Isodecanol (25339-17-7)
- M-Xylene (108-38-3)
- Methyl Oleate
- N-BenzylMethylAmine (103-67-3)
- N-Methylaniline (100-61-8)
- Ortho-Tolualdehyde (529-20-4)
- Propionic Anhydride
- Triisopropyl Phosphite
- Trimethyl Orthoformate
- Trimethoxyphenylsilane (2996-92-1)
- Ethyl Magnesium Bromide
- N,N,N,N-Tetramethyl-1,3-Propanediamine (110-95-2)
- Methanesulfonyl Chloride
- Dioxolane (646-06-0)
- O-Xylene (95-47-6)
- Crotonaldehyde (4170-30-3)
- 2-Butoxyethyl Acetate
- Methoxyacetyl Chloride
- Linear alkyl benzene
- 1,6-Dichlorohexane (2163-00-0)
- Methyl Soyate
- Sec-Butylbenzene (135-98-8)
- Cobalt Carbonate (513-79-1)
- Zinc Sulphide (1314-98-3)
- Zirconium Oxychloride (7699-43-6)
- Bismuth Trioxide (1304-76-3)
- 2-Amino-4-Chlorophenol (95-85-2) (C6H6ClNO)
- Borax Decahydrate (1303-96-4)
- Cadmium Oxide (1306-19-0)
- Boron (7440-42-8) (B)
- Calcium Bicarbonate (3983-19-5)
- Calcium Hydroxide (1305-62-0)
- Calcium Silicate (1344-95-2)
- 4-Dimethylaminopyridine (1122-58-3) (C7H10N2)
- Ammonium Tetrafluoroborate (13826-83-0)
- Aspartame (22839-47-0)
- Aluminium Isopropoxide (555-31-7)
- Dipotassium Phosphate Anhydrous
- Hydroxylamine Hydrochloride (5470-11-1)
- Hydroxypropyl Cellulose
- Monocalcium Phosphate Anhydrous
- N-Bromosuccinimide (0128-08-05) (C4H4BrNO2)
- N-Chlorosuccinimide (0128-09-06)
- Activated Alumina
- Alumina Hydrate
- Alumina Trihydrate
- Aluminium Hydroxide Trihydrate
- Aluminium Silicate
- Amorphous Silica
- Chromium Sulphate
- Colemanite
- Cerium Oxide
- Chloramine-T
- Cyclopentylmagnesium Bromide
- Diphenyl Isodecyl Phosphate
- Ethylene (74-85-1)
- French Chalk
- Potassium Cryolite
- Potassium Titanium Oxalate
- Lead Acetate Anhydrous
- Nickel Hydroxide
- Lithium Molybdate
- Lithium Silicate
- Lithium Tetraborate Anhydrous
- Magnesium Phosphate
- Para Hydroxy Benzoic Acid
- Maltodextrin (9050-36-6)
- Perlite (93763-70-3)
- Polyacrylamide (9003-05-08)
- Polyacrylic Acid
- Polydextrose (68424-04-04)
- Potassium Metabisulfite
- Potassium Oxalate
- Potassium Persulfate
- Potassium Phosphate
- Potassium Tetraborate Tetrahydrate
- Lead Carbonate
- Methyl Carbazate
- Mercuric Oxide Red
- Barium Titanate (12047-27-7)
- Bromocresol purple (115-40-2)
- 2-Methoxy-5-Nitroaniline (99-59-2) (C7H8N2O3)
- Barium Azide (18810-58-7)
- Ammonium Citrate Dibasic
- Potassium Thiocyanate
- Potassium Titanate
- Molybdenum Disulfide
- Molybdenum Trioxide
- Chromium Chloride
- Chromium Nitrate
- Chromium Oxide
- Boron Oxide
- Calcium Propionate
- Copper Sulphate Anhydrous
- Cyclopropanecarbonyl Chloride
- Dicyandiamide (461-58-5)
- Dextrose Anhydrous
- EDTA Dipotassium Salt
- EDTA Ferric Sodium
- Ferrous Sulfide
- Lead Tetroxide
- Litharge (1317-36-8)
- Manganese Dioxide
- Polysantol (107898-54-4)
- Potassium Bicarbonate
- Phosphogypsum Dihydrate
- P-Xylene (106-42-3)
- Poly(Diallyldimethylammonium Chloride)
- Aiocrol
- L+Tartaric Acid
- 2,4-Dinitrophenylhydrazine (119-26-6) (C6H6N4O4)
- Ammonium Pentaborate
- Hydrofluoric Acid 60%
- Hydrofluoric Acid 70%
- Benzyl Bromide
- Ferric Chloride Liquid
- Sodium Sulphide Hydrate Flakes
- Bentonite Powder
- Caustic Soda Lye
- Methyl Succinyl Chloride
- Aluminium Trihydroxide
- Phenyl Isocyanate
- Titanium Ethoxide
- Allyl Bromide
- Copal Gum
- Starch
- Phenyl (2396-01-02)
- Cationic Polyelectrolyte Powder
- Wacker L097
- Sodium Metatitanate
- Precipitated Silica
- Iron Oxide
- Red Phosphorus
- Silicon Carbide
- Alpha Arbutin
- Chromotropic Acid Disodium Salt
- Copper Pyrithione
- 4-Hydrazinobenzoic Acid (619-67-0)
- Ethylenediaminetetraacetic Acid Tripotassium Salt Dihydrate
- Roasted Molybdenum Concentrate
- Sodium Aluminate
- Selenium Dioxide
- Sodium Phosphate Tribasic Dodecahydrate
- Sodium Carboxymethyl Cellulose
- Sodium Caseinate
- Sodium Selenite
- Lithopone (1345-05-07)
- Gadolinium Oxide
- Kaolin (1332-58-7)
- Potassium Borohydride
- Quinizarin (81-64-1)
- Acetyl Acetone Peroxide
- Soda Lime
- Polyelectrolyte
- Arsenic Trioxide
- Safolite
- Zinc Borate
- N-Heptane 99%
- Kamicryl 400
- TCF-10
- Sunflower Acid oil
- 3,3'-Dichlorobenzidine (91-94-1)
- Sodium Bisulfate Monohydrate
- Oxone
- Formaldehyde Sodium Sulfoxylate Dihydrate
- Calcined Alumina
- PPG-2000
- 3-Bromoanisole (2398-37-0)
- LO Acid
- Lead Naphthenate
- White Spirit
- Tin Chloride Anhydrous
- Edta Calcium Disodium
- Heptanoic Anhydride
- Palladium on Activated Carbon 5%
- Bis-2-Ethylhexyl Sebacate
- 2,2-Dimethoxypropane (77-76-9)
- Polyglycerol Polyricinoleate
- EDTA Zinc
- Sodium Metavanadate
- Sodium Molybdate Anhydrous
- Sodium Oxalate
- Sodium Tungstate
- Stannic Oxide
- Strontium Chromate
- Thiourea Dioxide
- Strontium Sulphate
- Succinylcholine Chloride
- Titanium Oxychloride
- Vanadium Pentoxide
- Yttrium Oxide
- Tungstic Acid
- Zinc Chromate
- Zinc Formaldehyde Sulfoxylate
- Zinc Lactate
- Zinc Silicofluoride
- Polyaluminium Chloride
- Potassium Bisulfate
- Strontium Aluminate
- Potassium Periodate
- Potassium Fluorotitanate
- Pyrophyllite
- Silicon Nitride
- Sodium Bentonite
- Sodium Orthosilicate
- Tungsten Carbide
- Antimony Trisulphide
- Trimethylsulfoxonium Bromide
- Calcium Borate
- Cuprous Chloride
- Trisodium Citrate Anhydrous
- Sodium Tolyltriazole
- Hexafluorozirconic Acid
- 2,5-Dimethoxyaniline (102-56-7)
- 1,4-Dithiane-2,5-Diol (40018-26-6)
- Sodium Hypochlorite 5%
- Calcium Bentonite
- Potassium Tetrafluoroaluminate
- Glucose Syrup
- Alcohols C9-11-Iso- C10-Rich Ethoxylated
- Linseed Oil
- Chrome Oxide Green
- Mixed Xylene
- Bentonite (1302-78-9)
- Magnesium Acetate
- Sodium Formaldehyde Sulfoxylate
- Calcium Nitrite
- Sodium Stannate
- Aniline (62-53-3)
- N,N-Dimethyl aniline
- Allylamine (0107-11-9)
- Sulphur Monochloride
- 1-Ethoxy-2-Propanol (1569-02-04)
- Formaldehyde (50-00-0)
- Silicone Defoamer
- C12-C14 Alcohol ethoxylate 8 Mole Emulsifier
- Aluminium Sulphate Anhydrous
- Ammonium Lignosulfonate
- 2,2,2-Trichloroethanol (115-20-8)
- Benzeneseleninic Anhydride
- Calcite Powder
- Valeronitrile (110-59-8)
- Triethoxymethylsilane (2031-67-6)
- Ammonium Cerium Nitrate
- Isobutylene oxide
- Titanium Carbide
- Tellurium Dioxide
- Styrene (100-42-5)
- 3-Chloro-2-Methylaniline (87-60-5)
- Carboxymethyl Cellulose
- Potassium Peroxymonosulfate Triple Salt
- Sodium Acid Pyrophosphate
- 48-R
- Fumed Silica
- Spodumene Concentrate
- 2-Ethoxyethanol (110-80-5)
- Lawesson's Reagent
- 2-(2-Chloroethoxy)Ethanol (628-89-7)
- Tallow Amine Ethoxylate
- Methyl Silicate
- Dialdehyde Starch
- Bromine (7726-95-6)
- 2-Ethylhexanoic Acid
- 3-Chloro-4-Fluoroaniline (367-21-5)
- Barium sulfide
- Benzene (71-43-2) (C6H6)
- Nitromethane (75-52-5)
- Maleic Acid
- Chloroform (67-66-3)
- Decolite DP
- Tert-Butylhydroquinone (1948-33-0)
- Stearylamine (124-30-1)
- Stannous Fluoride
- Strontium Carbonate
- Triethylamine (121-44-8)
- Defoamer
- Methyl Linoleate
- 1-Naphthol (90-15-3) (C10H8O)
- 1,2-Epoxybutane (106-88-7)
- Butylated Hydroxytoluene
- Carbitol (111-90-0)
- Iodine Monochloride
- Hexamine (100-97-0)
- 1-Hydroxyethylidene-1,1-Diphosphonic Acid
- Monomethylamine (74-89-5)
- Lignin (94113-57-2)
- Nitroethane (79-24-3)
- Ferrous Chloride
- Para Nitrochlorobenzene
- Perchloroethylene (127-18-4)
- Phenidone (92-43-3)
- Phenol (108-95-2)
- Phosphorous Acid
- Methyl Cyanoacetate
- Potassium Bifluoride
- Methylal (109-87-5)
- Aluminum Sulfate Non Ferric
- Calcium Sulfate
- Diammonium Phosphate
- Dimethyl Sulphate
- EDTA Disodium Salt
- Ethyl Silicate
- Fluosilicic Acid
- Polyvinyl Alcohol
- Potassium Dichromate
- Potassium Silico Fluoride
- Sodium Borohydride
- Sodium Hydrosulfite
- Sodium Lignosulfonate
- Sodium sulfite
- Stannous Chloride
- Thiourea (62-56-6)
- Sulfamic Acid
- Toluene (108-88-3)
- Tert-Butyl Nitrite
- Triethyl Orthoformate
- Zeolites (1318-02-01)
- 1,1,2,2-Tetrachloroethane (79-34-5)
- Sodium Hypophosphite
- Sodium Hypophosphite
- Magnesium Silicofluoride
- 2,6-Dichlorobenzaldehyde (83-38-5)
- Calcium aluminate
- Calcium Lignosulphonate
- 4-Bromophenol (106-41-2)
- 4-Chlorobenzotrifluoride (98-56-6)
- 4-Tert-Butylcatechol (98-29-3)
- Acetone (67-64-1)
- Acrylic Acid
- Adipic Acid
- Barium Sulphate
- Aluminium Oxide
- Ammonium Oxalate
- Benzotriazole (95-14-7)
- N,N-Dimethylethanolamine (108-01-0)
- Nitrocellulose (9004-70-0)
- Maleic Anhydride
- Paraformaldehyde (30525-89-4)
- Phosphoric Acid
- Phosphorus Tribromide
- Poloxamer 407
- Potassium Hydrogen Phthalate
- Potassium Hydroxide
- Methyltrimethoxysilane (1185-55-3)
- Potassium Titanium Fluoride
- Propionitrile (107-12-0)
- Propionyl Chloride
- N-Propyl Acetate
- Propylene Glycol Monomethyl Ether Acetate
- 4-Methyl-2-Pentanol (0108-11-2)
- Aluminium Chloride
- Aluminium Trifluoride
- 3-AcetylPyridine (0350-03-08)
- Calcium Formate
- Tricalcium phosphate
- Copper Oxide
- Diethyl Ether
- Diethylene Glycol
- Diethylenetriamine (111-40-0)
- Disodium Phosphate Anhydrous
- DL-Tartaric Acid
- Furfuryl Alcohol
- Ethylenediamine (107-15-3)
- Glyoxal (107-22-2)
- Gum Rosin
- Ferric Sulfate
- Hydrobromic Acid
- Formamide (75-12-7)
- Potassium Benzoate
- Potassium Bromide
- Potassium Carbonate
- Potassium Chloride
- Methylcyclohexane (108-87-2)
- Potassium Ferricyanide
- Monoethanolamine (141-43-5)
- Potassium Fluoborate
- N-Methyl Pyrrolidone
- Propylene Carbonate
- Resist Salt
- Sodium Carbonate
- Sodium Benzoate
- Sodium Bicarbonate
- Sodium Persulfate
- Sodium Ferrocyanide
- Sodium Hypochlorite
- Sodium Metabisulphite
- Sodium Thiocyanate
- Tetrabutylammonium Bromide
- Tetraisopropyl Titanate
- Thiophenol (108-98-5)
- Sulfuric Acid
- Triacetin (102-76-1)
- Trichloroethylene (79-01-6)
- Tert-Butyl Chloride
- Trimethylamine (75-50-3)
- Lithium Hydroxide Monohydrate
- Methoxy Propanol
- N-Ethylethanamine (109-89-7) (C4H11N)
- Naphthalene (91-20-3) (C10H8)
- Tetrahydrofuran (109-99-9) (C4H8O)
- Sodium Hexametaphosphate
- Sodium Nitrite
- Sodium Metaborate
- Aluminum fluoride
- N-Butyl Bromide
- Sodium Hydroxide Flakes
- Sodium Petroleum Sulfonate
- Sodium Aluminum Silicate
- Dibasic Ester
- Personal & Home Care
- Citric Acid Anhydrous
- Cetyltrimethylammonium Bromide
- Alcohol Ethoxylate
- Aluminium Chlorohydrate
- Azelaic Acid
- Benzoyl Peroxide
- Calcium Thioglycolate
- Capric Acid
- Castor Oil
- Citric Acid Anhydrous
- Citric Acid Monohydrate
- Cocoamine
- Dehydroacetic Acid
- Denatonium Saccharide
- Dimethylpolysiloxane (9016-00-6)
- Emulsifying Wax
- Hexylene Glycol
- Hydrogen Peroxide
- Hydrogenated Castor oil
- Hydroquinone (123-31-9)
- Isopropyl Palmitate
- Lactic Acid
- Lanolin (8006-54-0)
- Lauryl Glucoside
- Lauryl Trimethyl Ammonium Chloride
- Methylparaben (99-76-3)
- Nonoxynol-9 (14409-72-4)
- Oleic Acid
- Oleyl Alcohol
- Olive Oil
- Palm Oil
- Palm Stearin
- Paraffin Wax
- Potassium Stearate
- Potassium Tripolyphosphate
- Propyl Gallate
- Propylene Glycol
- Propylparaben (94-13-3)
- Pyrithione Zinc
- Pyrithione Zinc
- Resorcinol (108-46-3)
- Rice Bran Wax
- Salicylic Acid
- Silicone Oil
- Sodium Percarbonate
- Tetrapotassium Pyrophosphate
- Tetrasodium Pyrophosphate Anhydrous
- 2-Ethylhexyl Laurate
- 2-Ethylhexyl Salicylate
- 3-O-Ethyl-l-Ascorbic Acid
- Alcohol Ethoxylates
- Allantoin (97-59-6)
- Ammonium Lactate
- Ammonium Thioglycolate
- Beeswax (8012-89-3)
- Behenyl Alcohol
- Benzalkonium Chloride
- Benzethonium Chloride
- Benzyl Nicotinate
- Beta Glucan
- Bismuth Oxychloride
- Bronopol (52-51-7)
- Butylated Hydroxyanisole
- Calamine (8011-96-9)
- Calcium Hypochlorite
- Candelilla Wax
- Caprylyl Glycol
- Cetrimide (8044-71-1)
- Cetyl Alcohol
- Cetylpyridinium Chloride
- Climbazole (38083-17-9)
- Coco Monoethanolamide
- Coconut Oil
- Decyl Alcohol
- Decyl Glucoside
- Diethyl Phthalate
- Dihydroxyacetone (96-26-4)
- Dimethyl Isosorbide
- Dodecyl Benzene
- Dodecyl Sulfate Sodium Salt
- Dodecylbenzene Sulphonic Acid
- Dodecylbenzenesulfonic acid
- Ethylhexyl Oleate
- Glycerin (56-81-5)
- Hydroxypropyl Methyl Cellulose
- Glycerol Monolaurate
- Glycolic Acid
- Hydrogenated Palm Stearin
- Hydroxyethyl Cellulose
- Isododecane (31807-55-3)
- Isopropyl Myristate
- Isothiazolinone (1003-07-02)
- Kojic Acid
- L-Ascorbyl Palmitate
- Lactic Acid 90%
- Lauric Acid
- Lauric Acid Diethanolamide
- Linear Alkyl Benzene Sulfonic Acid
- Linoleic Acid
- Lutensol XL 80
- Magnesium Aluminium Silicate
- Methyl Hydrogenated Rosinate
- Microcrystalline Wax
- Mineral Oil
- Myristyl Alcohol
- Octyl Stearate
- Palmitic Acid
- Paraffin Oil
- Peracetic Acid
- Polyquaternium 47
- Polysorbate 20
- Potassium Oleate
- Propylene Glycol Monostearate
- Rice Bran Wax Pastilles
- Soap Noodles
- Sodium Dichloroisocyanurate
- Sodium Lauryl Ether Sulphate
- Sodium levulinate
- Sodium Metasilicate
- Sodium Monofluorophosphate
- Sodium Oleate
- Sodium Picramate
- Sodium Stearate
- Sodium Tripolyphosphate
- Sodium xylenesulfonate
- Sorbitan Monooleate
- Sorbitan Monostearate
- Sorbitan Trioleate
- Sorbitol (50-70-4)
- Soya Wax
- Spilanthol 20%
- Stearic Acid
- Stearyl Alcohol
- Talcum (14807-96-6)
- Tetraacetylethylenediamine (10543-57-4)
- Thioglycolic Acid
- Titanium Dioxide
- Titanium Dioxide Rutile 902
- Tributyl Citrate
- Triethanolamine 99%
- Trimethylsiloxysilicate (56275-01-05)
- Trisodium Phosphate Anhydrous
- Triton X-100
- Urea Hydrogen Peroxide
- Xanthan Gum
- Zeolite 4A
- Zinc Ricinoleate
- 2-Ethylhexyl 4-Aminobenzoate
- 3-[(2-Ethylhexyl)Oxy]-1,2-Propanediol (70445-33-9)
- Aloe Vera
- Ammonium Lauryl Sulfate
- Benzisothiazolinone (2634-33-5)
- Beta-Carotene
- Caprylic/Capric Triglyceride
- Carnauba Wax
- Ceresin Wax
- Cetostearyl Alcohol
- Cetrimonium Chloride
- Chloroxylenol (88-04-0)
- Cocamide DEA
- Cocamidopropyl Betaine
- Coconut Fatty Acid
- D-Panthenol (81-13-0)
- Didecyldimethylammonium Chloride
- Dipropylene Glycol Monomethyl Ether
- Disodium Lauryl Sulfosuccinate
- Hydrogenated Palm Oil
- Iodophor
- Macrogolglycerol Ricinoleate
- Niacinamide Ascorbate
- Nonylphenol Ethoxylate 9.5 Mole
- Octyl Methoxycinnamate
- Sodium Cocoyl Isethionate
- Sodium Dl-Lactate
- Sodium Dodecylbenzenesulfonate
- Sodium Lauroyl Sarcosinate
- Sodium Methylparaben
- Sodium Perborate Anhydrous
- Sodium Perborate Tetrahydrate
- Sodium Propylparaben
- Sorbic Acid
- Sorbitan Tristearate
- Trichloroisocyanuric Acid
- Triclosan (3380-34-5)
- Tween 20
- TWEEN 60
- Tween 80
- 1,3-Butanediol (107-88-0)
- Aluminum Chlorohydrate Dihydrate
- Hyaluronic Acid
- N-Acetyl-D-Glucosamine
- Baking Powder
- Disodium Laureth Sulfosuccinate
- Fatty Alcohol Ethoxylate
- Glycerol Alpha-Monostearate
- Hydrogenated Soybean Oil
- Peanut Oil
- Petroleum Jelly
- Polyquaternium-7 (26590-05-6)
- Silicone Emulsion
- Sodium Bromate
- Sodium Chlorite 80%
- Xylitol (87-99-0)
- Zinc disodium EDTA 15%
- P-Phenylenediamine (106-50-3)
- Agrochemicals
- Zinc Sulphate Anhydrous
- 2-Chlorobenzonitrile (873-32-5)
- Cuelure (3572-06-03)
- Sodium Nitrate
- Azadirachtin (11141-17-6)
- Pyrethrum (8003-34-7)
- Potassium Sulphate
- Potassium Silicate
- Potassium Nitrate
- Magnesium Sulphate Anhydrous
- Ammonium Sulfamate
- Diatomaceous Earth
- 1,2-Dibromoethane (106-93-4)
- Zinc Sulphate Heptahydrate
- 2,6-Difluorobenzamide (18063-03-01)
- 1,2,4-Triazole (288-88-0)
- Bromadiolone (28772-56-7)
- 2,6-Dichlorobenzonitrile (1194-65-6)
- Neem Oil
- Calcium Cyanamide
- Liquor Ammonia
- Organic Manure
- Calcium Nitrate
- Calcium Peroxide
- Monoammonium Phosphate
- Acetoacet Ortho Chloroanilide
- Rock Phosphate
- Cyanamide (0420-04-02)
- Tributyltin Oxide
- Copper Oxychloride
- Urea (57-13-6)
- Zinc Phosphide
- Triacontanol (593-50-0)
- 1-Naphthaleneacetic Acid
- Temephos (3383-96-8)
- Gibberellic Acid
- Sulfur (7704-34-9)
- Sodium Pentachlorophenate
- Sodium Humate
- Potassium Humate
- Potassium Diformate
- Fulvic Acid
- Humic Acid
- Disodium Octaborate Tetrahydrate
- Cyromazine (66215-27-8)
- Custard Apple Seed Extract
- Butaphosphan
- 2,4,6-Trichlorophenol (88-06-2)
- Propoxur (114-26-1)
- Manganese Sulphate Anhydrous
- Manganese Carbonate
- Paraquat Dichloride
- Malathion (121-75-5)
- Magnesium Nitrate Hexahydrate
- Nicotine Sulphate
- Mono Potassium Phosphate
- Hexaconazole (79983-71-4)
- Ammonium Sulfate
- Ammonia (7664-41-7)
- Amitraz (33089-61-1)
- Plastic & Rubber Chemicals
- Zinc Stearate
- Calcium Stearate
- Alpha-Methylstyrene (98-83-9)
- Decabromodiphenyl Ethane
- Zinc Oxide
- Zinc Hydroxide
- Triphenyl Phosphite
- Triphenyl Phosphate
- Ammonium Polyphosphate
- Ammonium Stearate
- Tricresyl Phosphate
- Toluene Diisocyanate
- Tetrabromobisphenol A
- 1,4-Butanediol (110-63-4)
- 2-Acrylamido-2-Methylpropane Sulfonic Acid
- 3-Glycidyloxypropyltrimethoxysilane
- Polyester Resins
- Triallyl Isocyanurate
- Triallyl Cyanurate
- Cellulose Acetate
- Pigment Red 4
- Cobalt Octoate
- Bisphenol A
- 2-Ethylhexyl Acrylate
- 2-Hydroxyethyl Acrylate
- Polyethylene Wax
- Caprolactam (105-60-2)
- 2-Mercaptobenzothiazole (149-30-4)
- Ketonic Resin
- Polyamide resin
- Cellulose Acetate Butyrate
- Divinylbenzene (1321-74-0)
- Polytetramethylene Ether Glycol
- Phenol Formaldehyde Resin
- N-Tert-Butylacrylamide (107-58-4)
- Epoxy Resin
- Montan Wax
- Chlorinated Polyethylene
- Diphenylguanidine (0102-06-07)
- Hydroxypropyl Acrylate
- Acrylamide (79-06-1)
- Azodicarbonamide (123-77-3)
- Polypropylene (9003-07-0)
- N-Acetylcaprolactam (1888-91-1)
- Aluminium Stearate
- Benzoguanamine (91-76-9)
- Benzothiazole (95-16-9)
- Trimellitic Anhydride
- Trimellitic Anhydride
- Polylactic Acid
- Redispersible Polymer
- Dodecanedioic Acid
- Dimethyl Terephthalate
- Diacetone Acrylamide
- Dicyclopentadiene (77-73-6)
- Phthalic Anhydride
- P-Toluenesulfonamide (70-55-3)
- N,N'-Methylenebisacrylamide (110-26-9)
- O-Toluenesulfonamide (88-19-7)
- Trimethylolpropane (77-99-6)
- Triethylenediamine (280-57-9)
- Tert-Butyl Peroxybenzoate
- Strontium Hydroxide
- Sodium Polyacrylate
- Benzyl Butyl Phthalate
- Para-Tertiary Butylphenol
- Polyvinyl Chloride
- Polyvinyl Butyral
- Polyvinyl Acetate
- Polyurethane
- Polymethyl Methacrylate
- Lead Stearate
- Ethylene Vinyl Acetate
- Glycidyl Methacrylate
- Ethylene Bisstearamide
- Dipropylene Glycol Dibenzoate
- Erucamide (112-84-5)
- Dipentaerythritol (126-58-9)
- Dioctyl Adipate
- Dimethyl Adipate
- Dimer Acid
- Isophthalic Acid
- Diethylene Glycol Dibenzoate
- Dicumyl Peroxide
- Dibutyltin Dilaurate
- Chlorinated Paraffin Wax
- Cyclohexanone Formaldehyde Resin
- Antimony Trioxide
- Itaconic Acid
- Tin 2-Ethylhexanoate
- Polyethylene Glycol Diacrylate
- Retarder
- Di-Tert-Butyl Peroxide
- Benzylamine (100-46-9)
- Dodecyl Mercaptan
- Dioctyl Phthalate
- Dioctyl Maleate
- Diallyl Phthalate
- Diallyl Maleate
- Diisononyl Phthalate
- Dicyclohexyl Phthalate
- Dibutyl Sebacate
- Dibutyl Phthalate
- Dibutyl Maleate
- Dibutyl Adipate
- Ammonium Polyacrylate
- 3-Aminopropyltrimethoxysilane (13822-56-5)
- 3-Aminopropyltriethoxysilane (919-30-2)
- Aluminium Trihydrate
- Acrylonitrile (107-13-1)
- Propylene Oxide
- 3-Trimethoxysilyl propyl methacrylate
- Methyl Methacrylate
- Polyethylene (9002-88-4)
- Bis-2-Propylheptyl Phthalate
- Urea Formaldehyde
- Polybutylene Succinate
- Polydimethylsiloxane Vinyl Terminated
- Silicone Resin
- Allyl Glycidyl Ether
- 3-Mercaptopropyl Trimethoxysilane
- Acrylic Emulsion
- Acrylic Thickener
- PBR CISAMER 01
- Polypropylene Glycol
- Barium Stearate
- Allyl Methacrylate
- Hydroxyethyl Methacrylate
- Diisodecyl Adipate
- Butyl Acrylate
- 4-Tert-Octylphenol (140-66-9)
- Vinyl Acetate Monomer
- Trimethylolpropane Triacrylate
- Hexamethylenediamine (0124-09-04)
- Methyl Acrylate
- Methacrylic Acid
- Methacrylamide (79-39-0)
- Melamine Cyanurate
- Melamine (108-78-1)
- Pentaerythritol (115-77-5)
- Oleamide (301-02-0)
- Neopentyl Glycol
- N-Octyl Mercaptan
- Pharma Chemicals
- Clonidine Related Compound A
- Zinc Picolinate
- Zoledronic Acid
- Bismuth Subnitrate
- Bismuth Subsalicylate
- 2-Amino-5-Chlorobenzophenone (719-59-5)
- 2-Aminomethyl-1-Ethylpyrrolidine (26116-12-1)
- Busulfan (55-98-1)
- 2-Mercapto-5-Methoxybenzimidazole (37052-78-1)
- Caffeine (58-08-2)
- Caffeine Citrate
- Calcium Bisglycinate
- Calcium D-Saccharate
- Calcium Gluconate
- Calcium Glycerophosphate
- 4-Aminobenzoic Acid
- 4-Chlorosalicylic Acid
- Canagliflozin Hemihydrate
- Candesartan Cilexetil
- Carbamazepine (298-46-4)
- Carbidopa (28860-95-9)
- Activated Charcoal
- Acyclovir (59277-89-3)
- Adapalene (106685-40-9)
- Carprofen (53716-49-7)
- Carvedilol (72956-09-03)
- Celecoxib (169590-42-5)
- Cellulose Acetate Phthalate
- Cetirizine Dihydrochloride
- Citicoline Sodium
- 5-Chlorosalicylic Acid
- Clindamycin Phosphate
- Clomiphene Citrate
- Clonidine Hydrochloride
- Clopidogrel Bisulfate
- 5-Sulphosalicylic Acid
- Clotrimazole (23593-75-1)
- Abiraterone Acetate
- Amodiaquine Hydrochloride
- Ascomycin (104987-12-4)
- Acetylsalicylic Acid
- Acriflavine Hydrochloride
- Astaxanthin (472-61-7)
- Azithromycin Anhydrous
- Baclofen (1134-47-0)
- Barbituric Acid
- Albendazole (54965-21-8)
- Benfotiamine (22457-89-2)
- Benzocaine (94-09-7)
- Benzoic Acid
- Indomethacin (53-86-1)
- Inulin (9005-80-5)
- Iodine (7553-56-2)
- Ipriflavone (35212-22-7)
- Beta Cyclodextrin
- Isonicotinamide (1453-82-3)
- Betamethasone Dipropionate
- Biotin (58-85-5)
- Docusate Sodium
- Itraconazole (84625-61-6)
- Ivermectin (70288-86-7)
- Ketoprofen (22071-15-4)
- L-Arginine Alpha-Ketoglutarate
- L-Arginine L-Aspartate
- l-Arginine Monohydrochloride
- L-Carnitine (541-15-1)
- Hydrochlorothiazide (58-93-5)
- Hydrocortisone (50-23-7)
- L-Carnosine (305-84-0)
- Hydroxychloroquine Sulfate
- L-Cysteine (52-90-4)
- Hydroxyethyl Starch
- L-Cystine (56-89-3)
- L-Glutamine (56-85-9)
- Ibuprofen (15687-27-1)
- L-Histidine (71-00-1)
- Hydroxyprogesterone Caproate
- L-Leucine (61-90-5)
- L-Mandelic Acid
- L-Methionine (63-68-3)
- L-Phenylalanine (63-91-2)
- L-Pyroglutamic Acid
- Montelukast Sodium
- Mupirocin (12650-69-0)
- L-Threonine (72-19-5)
- L-Tryptophan (73-22-3)
- L-Valine (72-18-4)
- N-Butyl Cyanoacrylate
- Lactitol Anhydrous
- Lansoprazole (103577-45-3)
- Lanthanum Carbonate
- Nabumetone (42924-53-8)
- Alfuzosin Hydrochloride
- Alprostadil (745-65-3)
- L-Selenomethionine (3211-76-5)
- N,N-Dicyclohexylcarbodiimide (538-75-0)
- Nalidixic Acid
- Naltrexone Hydrochloride
- Nebivolol (99200-09-06)
- Leflunomide (75706-12-6)
- Lenalidomide (191732-72-6)
- Niacin (59-67-6)
- Nicardipine Hydrochloride
- Letrozole (112809-51-5)
- Levodopa (59-92-7)
- Levosulpiride (23672-07-03)
- Lidocaine (137-58-6)
- Niclosamide (50-65-7)
- Nicotinamide (98-92-0)
- Nitazoxanide (55981-09-04)
- Locust Bean Gum
- Loperamide Hydrochloride
- Lumefantrine (82186-77-4)
- Magnesium Acetyl Taurate
- Olmesartan Medoxomil
- Magnesium Citrate
- Ondansetron (99614-02-05)
- Orlistat (96829-58-2)
- Magnesium Hydroxide
- Orphenadrine Citrate
- Magnesium Lactate Anhydrous
- Oxybutynin Chloride
- Magnesium orotate
- Magnesium Pidolate
- Pantoprazole Sodium
- Para-Hydroxyphenylacetic Acid
- Mandelic Acid
- Manganese Gluconate
- Meclizine (569-65-3)
- Meglumine (6284-40-8)
- Phenothiazine (92-84-2)
- Metformin Hydrochloride
- Phenylbutazone (50-33-9)
- Methylsulfonylmethane (67-71-0)
- Metoclopramide (364-62-5)
- Pralidoxime Iodide
- Pramoxine Hydrochloride
- Pravastatin Sodium
- Miglustat (72599-27-0)
- Pregabalin (148553-50-8)
- Progesterone (57-83-0)
- Minoxidil (38304-91-5)
- 4-Methoxy Phenylacetic Acid
- Pyridostigmine Bromide
- Pyrimethamine (58-14-0)
- Alendronate Sodium
- 2-Phenylethylamine (64-04-0)
- Nitrofurantoin (67-20-9)
- Oxiconazole Nitrate
- Methotrexate (59-05-2)
- Methylprednisolone Aceponate
- Amino Acid
- Chlortetracycline
- Chlorthalidone (77-36-1)
- Choline Chloride
- Choline Dihydrogen Citrate
- Choline Salicylate
- Bismuth Carbonate
- Chromium Picolinate
- Calcium Ascorbate
- Calcium Folinate
- Calcium Hydrogen Phosphate
- Copper Gluconate
- Calcium Phosphoryl Choline Chloride
- Croscarmellose Sodium
- Chloramphenicol (56-75-7)
- Chlorhexidine Gluconate
- Chlorobutanol (57-15-8)
- Cyproheptadine Hydrochloride
- Cytidylic Acid
- Dicaffeine Malate
- Diclofenac Diethylamine
- Dextran (9004-54-0)
- Diethylamine Salicylate
- Dextromethorphan (125-71-3)
- D-Galactose (59-23-4)
- Dibenzoyl-D-Tartaric Acid
- Dimethyl Fumarate
- D-Xylose (58-86-6)
- Econazole Nitrate
- Enrofloxacin (93106-60-6)
- Dimethylglycine (1118-68-9)
- Diphenhydramine Hydrochloride
- Erythritol (149-32-6)
- Doxycycline Hydrochloride
- Doxycycline Monohydrate
- Fructose (57-48-7)
- Gatifloxacin Sesquihydrate
- Gliclazide (21187-98-4)
- Glimepiride (93479-97-1)
- Glucosamine Hydrochloride
- Glycine (56-40-6)
- Ethylenediamine Dihydroiodide
- Famotidine (76824-35-6)
- Fenofibrate (49562-28-9)
- Ferric Ammonium Citrate
- Ferrous Ascorbate
- Ferrous Bisglycinate
- Inositol (87-89-8)
- Ferrous Gluconate
- L-Aspartic Acid
- Flurbiprofen (5104-49-4)
- Folic Acid
- Magnesium Gluconate
- Maize Starch
- Nimbin (5945-86-8)
- Adenosylcobalamin (13870-90-1)
- Chlorogenic Acid
- Colchicine (64-86-8)
- Dasatinib (302962-49-8)
- Dimenhydrinate (523-87-5)
- Dibenzoyl-L-Tartaric Acid
- Estradiol Anhydrous
- Doxorubicin Hydrochloride
- Furosemide (54-31-9)
- Gamma-Aminobutyric Acid
- Glibenclamide (10238-21-8)
- Ezetimibe (163222-33-1)
- Polyvinylpyrrolidone (9003-39-8)
- Methylcobalamin (13422-55-4)
- Methylene Blue
- Quinine Sulphate
- Pectin (9000-69-5)
- Resveratrol (501-36-0)
- Risperidone (106266-06-2)
- Saccharin (81-07-2)
- Selenious Acid
- Serine (56-45-1)
- Sodium Stearyl Fumarate
- Tetraethylammonium Bromide
- Thiamine Hydrochloride
- Strontium Ranelate
- Strychnine Sulphate
- Titanium Diboride
- Sulphamethoxazole (723-46-6)
- Tranexamic Acid
- Taurine (107-35-7)
- Trientine (112-24-3)
- Valproic Acid
- Vinpocetine (42971-09-05)
- Vitamin B12
- Vitamin D3
- Tripotassium Citrate
- Triprolidine Hydrochloride
- Atovaquone (95233-18-4)
- Curcumin (458-37-7)
- Fenbendazole (43210-67-9)
- L-Lysine (56-87-1)
- N-Acetyl L-Cysteine
- Octenidine Hydrochloride
- Piperidine (110-89-4)
- Policosanol (142583-61-7)
- Pyridoxine Hydrochloride
- Riboflavin (83-88-5)
- Terbutaline Sulfate
- Monosodium Citrate
- Di-Tert-Butyl Dicarbonate
- Quinine (130-95-0)
- Sodium Glycerophosphate
- Potassium Alginate
- Carrageenan (9000-07-01)
- Halquinol (8067-69-4)
- Magnesium L-Aspartate
- Povidone Iodine
- Sodium Polystyrene Sulfonate
- 2-1-Cyclohexenyl Ethylamine
- Sodium Saccharin
- Phosphatidylserine (51446-62-9)
- Sodium Potassium Tartrate
- Di-P-Toluoyl-L-Tartaric Acid Monohydrate
- Thiamine
- High Fructose Corn Syrup
- Larch Turpentine Oil
- Magnesium Hydroxide Ip
- Econazole Nitrate Ip
- Sodium Iodide BP
- Calcium Lactate Gluconate
- Ferrous Fumarate
- Thiopental Sodium
- Policosanol Extract
- Domperidone IP
- Vitamin D2
- Calcium Citrate Malate
- Vitamin A
- L-Theanine (3081-61-6)
- L-Lysine Hydrochloride
- 2-Acetylbutyrolactone (517-23-7)
- N-Hydroxy Succinimide
- Aluminum Acetate
- N,N-Diisopropylcarbodiimide (693-13-0)
- Acethydrazide (1068-57-1)
- Tricalcium Phosphate IP
- Adenine (73-24-5)
- Corn Syrup
- 5-Amino-6-methylbenzimidazolone
- Agar-Agar (9002-18-0)
- Dextromethorphan Hydrobromide
- Betaine Anhydrous
- Boric Acid IP
- Homoveratrylamine (120-20-7)
- Bismuth Subgallate
- Magnesium-L-Threonate (778571-57-6)
- Erdosteine (84611-23-4)
- Trimethyl Chitosan
- Rabeprazole Sodium
- Nicotine Benzoate
- Mannitol (69-65-8)
- Linolenic Acid
- Dextrose Monohydrate
- Ciprofloxacin Hydrochloride
- Uracil (66-22-8)
- Tyramine Hydrochloride
- Zinc Monomethionine
- Zinc Gluconate
- Trospium Chloride
- Zinc Aspartate
- Tert-Butyl Bromoacetate
- Terbinafine Hydrochloride
- Tenofovir Disoproxil Fumarate
- Tadalafil (171596-29-5)
- Tizanidine Hydrochloride
- Tetracaine Hydrochloride
- Sodium Butyrate
- Sildenafil Citrate
- Sodium Alginate
- Salicylamide (65-45-2)
- Simethicone (8050-81-5)
- Quinine Hydrochloride
- Potato Starch
- Diacerein (13739-02-01)
- Dehydrocholic Acid
- Entecavir Anhydrous
- Epirubicin Hydrochloride
- Diosmin (520-27-4)
- Erythorbic Acid
- Acesulfame Potassium
- Eszopiclone (138729-47-2)
- Ethacridine Lactate
- Dithranol (1143-38-0)
- Dopamine Hydrochloride
- Fusidic Acid
- Gefitinib (184475-35-2)
- Glycyrrhizin (1405-86-3)
- Fenticonazole Nitrate
- Ferric Carboxymaltose
- Finasteride (98319-26-7)
- Fluphenazine Hydrochloride
- Polynoxylin (9011-05-06)
- Pyrroloquinoline Quinone
- Racecadotril (81110-73-8)
- Reserpine (50-55-5)
- Ritonavir (155213-67-5)
- Rosmarinic Acid
- Roxithromycin (80214-83-1)
- Simvastatin (79902-63-9)
- Sodium Aminosalicylate
- Sertaconazole Nitrate
- Sevelamer Carbonate
- Tetracycline Hydrochloride
- Sugammadex Sodium
- Tolvaptan (150683-30-0)
- Trandolapril (87679-37-6)
- Tamoxifen Citrate
- Tamoxifen Citrate
- Merbromin
- Canola Oil
- Ascorbic Acid
- Ammonium Acetate
- Fish Oil
- Tacrolimus
- Paclitaxel IP
- L-Thyroxine
- Zinc Citrate
- Tannic Acid
- D-Pantothenic Acid Hemicalcium Salt
- Gelatin
- Lignocaine Hydrochloride
- 3,4-Diaminobenzoic acid
- Nicotine Salicylate
- Pentadecanoic Acid
- Oxytetracycline Hydrochloride
- Triphosgene
- Trimethoprim
- Tributyrin
- Sucralose
- Theobromine
- Stannous Pyrophosphate
- Sodium Ascorbate
- Potassium Gluconate
- Potassium Bitartrate
- Free Flow Salt
- Ferrous Sulphate Anhydrous
- Guar Gum
- Glucono Delta Lactone
- Gluconic Acid
- L-Arginine
- Imidazole
- Monosodium Glutamate Anhydrous
- Lactose Anhydrous
- Lafutidine
- Nicotine
- L-Glutathione
- L-Proline
- Omeprazole
- Paracetamol
- Potassium Iodate
- Potassium Iodide
- Potassium Sorbate
- Miconazole Nitrate
- Microcrystalline Cellulose
- Rapeseed Oil
- 2,4-Dihydroxybenzoic Acid
- Chloral Hydrate
- Cyclohexylamine
- Cysteamine Hydrochloride
- Diclofenac Sodium
- Degarelix Acetate
- Dextrin
- Dimethyl Sulfoxide
- Disulfiram
- D-Mandelic Acid
- D-Tartaric Acid
- Aroma Chemicals
- Home Page
- Company Profile
- Contact Us

- Home
- Products
- Industrial Chemicals
- Sodium Bifluoride
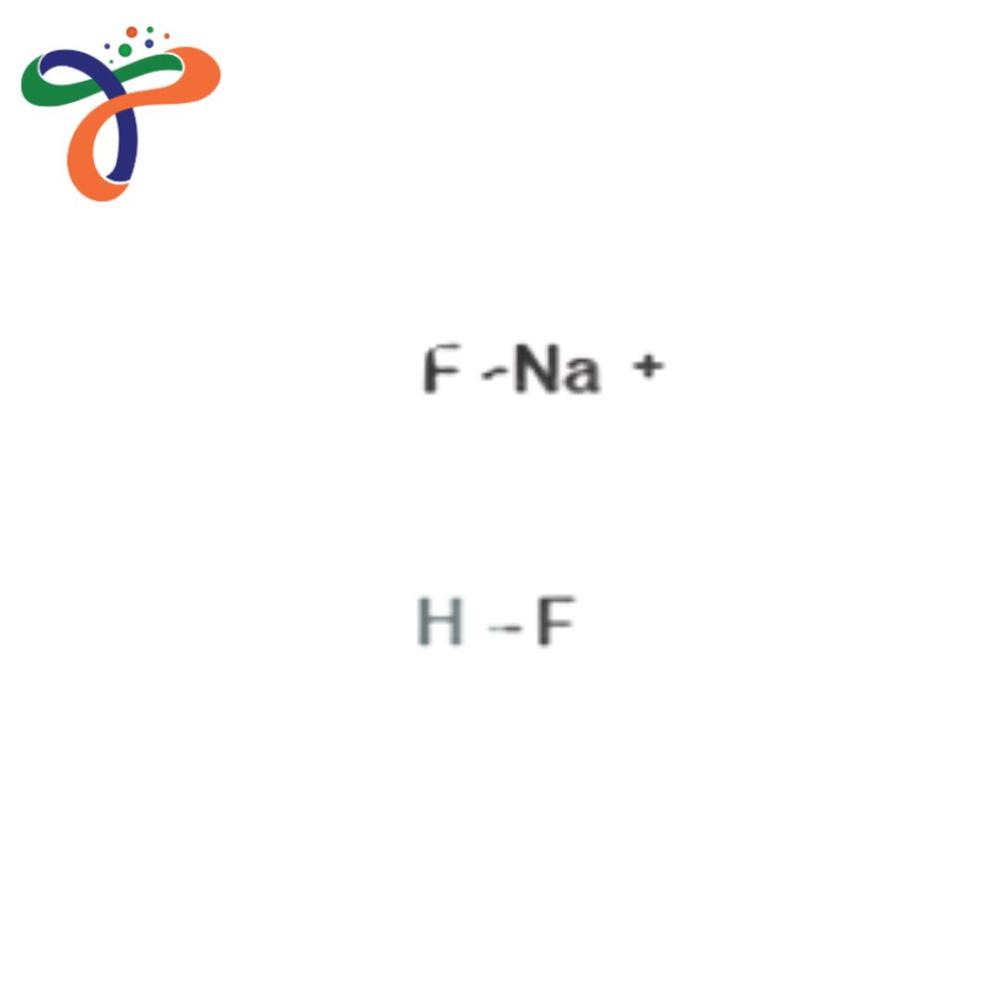
Sodium Bifluoride
Price 110.0 INR/ Kilograms
MOQ : 1 Kilograms
Sodium Bifluoride Specification
- Properties
- sodium bifluoride is highly soluble in water, producing acidic solutions due to hydrolysis. Industrially, sodium bifluoride is used in glass etching, metal cleaning, and surface treatment because of its ability to react with silica and oxides
- Melting Point
- 236 C
- Application
- Industrial
- Solubility
- Soluble in water (42 g/100 mL at 25 C); insoluble in alcohol
- CAS No
- 1333-83-1
- Usage
- Sodium Bifluoride Is A Highly Reactive Inorganic Compound Widely Used Across Industrial And Chemical Processing Applications. It Is Commonly Used In Metal Surface Treatment For Cleaning, Rust Removal, And Preparation Before Electroplating.
- Molecular Formula
- NaHF
- Appearance
- White crystalline solid
- Molecular Weight
- 61.99 g/mol Grams (g)
- Physical Form
- Solid
About Sodium Bifluoride
Sodium Bifluoride Is A Highly Reactive Inorganic Compound Widely Used Across Industrial And Chemical Processing Applications. It Is Commonly Used In Metal Surface Treatment For Cleaning, Rust Removal, And Preparation Before Electroplating.Efficient Surface Treatment Solution
Sodium bifluoride acts as a powerful agent for treating and cleaning metal surfaces. Its capability to remove oxides and rust, along with deep cleaning, makes it indispensable before processes such as electroplating. This ensures optimal performance and durability in finished products used across various industries.
Versatile Application Across Industries
This highly reactive inorganic compound finds extensive use in glass manufacturing and chemical processing. Its solubility in water and ability to interact with silica provides advantages for glass etching and industrial cleaning, making it an industry standard for precision and reliability in surface treatment.
FAQ's of Sodium Bifluoride:
Q: How is sodium bifluoride used in industrial metal cleaning and surface preparation?
A: Sodium bifluoride is applied to metal surfaces to clean them and remove rust or oxide layers. This process prepares metals for subsequent electroplating or other finishing steps by ensuring a contaminant-free surface.Q: What benefits does sodium bifluoride offer in glass etching and industrial cleaning processes?
A: Its high reactivity with silica and oxides enables efficient etching and cleaning, resulting in smooth, impurity-free surfaces. This enhances the quality and longevity of industrial glass items and metal products.Q: When should sodium bifluoride be used for metal surface treatment?
A: It is best used prior to electroplating or painting, during the preparation stage, to guarantee rust and oxide removal-thereby ensuring superior adhesion and a flawless finish.Q: Where is sodium bifluoride typically applied in industrial settings?
A: Sodium bifluoride is commonly utilized in plants specializing in glass manufacturing, chemical processing, and metal treatment. Major users include industries that require precision cleaning, like electronics, automotive, and construction sectors.Q: What is the manufacturing process for sodium bifluoride?
A: Sodium bifluoride is generally produced by reacting sodium fluoride with hydrofluoric acid. Careful control ensures purity and solid crystalline formation suitable for industrial application.Q: What is the molecular formula and CAS number of sodium bifluoride?
A: The molecular formula is NaHF, and it is registered under CAS No. 1333-83-1.

Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Name
Comapny Name
Phone Number
Email Id
City / State
Confirm Your Requirement
Verification Code
Did not receive yet?
Resend OTP

You’re Done!
We have received your requirements and will reply shortly with the best price.
Products You May Like
More Products in Industrial Chemicals Category



X
×
Add a mobile number to receive call from "TRIVENI INTERCHEM PVT. LTD."
×
Enter OTP
×
Share additional details for a quick response
×
Thank You!
Thank You for your valuable time. We have received your details and will get back to you shortly.
Our Products
134, Pancharatna, Char Rasta, GIDC, Vapi - 396195, Gujarat, India

Mr Rohit Ranjan
(VP)
Mobile :08045816834
TRIVENI INTERCHEM PVT. LTD.
All Rights Reserved.(Terms of Use)
Developed and Managed by Infocom Network Private Limited.
Developed and Managed by Infocom Network Private Limited.

 Send Inquiry
Send Inquiry



 Send Inquiry
Send Inquiry Call Me Free
Call Me Free English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese