-
Our Products
- Aroma Chemicals
- Citronellyl Tiglate
- Cis-3 Hexenyl Butyarate
- Cis-3-Hexenyl Methyl Carbonate
- Di Methyl Octanyl Acetate
- Cyprene Ex Cypriol
- Citronellyl Propionate
- Cis-3-Hexenyl Acetate (3681-71-8)
- Citronellyl Butyrate
- Cis-3 Hexenyl Caproate
- Cis-3-Hexenyl Benzoate (25152-85-6)
- Dimethyl Benzyl Carbinol
- Citronellyl Isobutyrate (97-89-2)
- Cinnamic Aldehyde (104-55-2)
- Dimethyl Disulfide (624-92-0)
- Isoamyl Salicylate
- Ethyl Laurate (106-33-2)
- Ethyl Benzoate (93-89-0)
- Hydratropic Aldehyde (93-53-8)
- Ethyl Lactate (97-64-3)
- Ethyl Maltol (4940-11-8)
- Lauryl Alcohol (112-53-8)
- Methyl Octanoate (0111-11-5)
- Gum Arabic (9000-01-05)
- Ethyl Acrylate (140-88-5)
- Hydratropic Alcohol
- Menthyl Acetate
- Ethyl Anthranilate
- Ethyl Formate (109-94-4)
- Methyl 4-Methyl Benzoate
- Iso Amyl Iso Valerate
- Iso Amyl Butyrate
- Isopropyl Acetate (108-21-4)
- Sandalwood Oil (8006-87-9)
- N-Butyl Anthranilate
- Iso Amyl Phenyl Acetate
- Iso Butyl Phenyl Acetate
- Mint Terpenes
- Methyl Propionate
- 2-Acetyl Furan (1192-62-7)
- Para Cresyl Acetate
- Pomelo Acetal
- Methyl Propyl Ketone
- Isobutyl Isobutyrate (97-85-8)
- Mesityl Oxide (141-79-7)
- L-Glutamic Acid (56-86-0)
- Methyl Chemical
- Isopropenyl Acetate
- Iso Propyl Propionate
- Styrallyl Propionate
- Methyl 2-Methyl Benzoate
- Methyl Octine Carbonate
- Isoamyl Benzoate (94-46-2)
- Isopropyl Phenyl Butanal
- Yeast Extract (8013-01-2)
- Isobornyl Cyclohexanol (3407-42-9)
- Muguet Aldehyde
- Mushroom Alcohol (3391-86-4)
- Styrallyl Alcohol
- Methyl Salicylate (119-36-8)
- Phenyl Ethyl Cinnamate
- Vetiveryl Acetate (62563-80-8)
- Neo Menthol (2216-52-6)
- Phenoxy Ethyl Isobutyrate
- Phenyl Ethyl Formate
- Peony Acetonitrile
- Terpinyl Acetate
- N-Caproic Acid
- Phenyl Ethyl Isobutyrate
- Di Methyl Octanol
- Phenyl Ethyl Tiglate
- Phenylethyl Bromide (103-63-9)
- N-Propyl Acetate
- Phenyl Ethyl Phenyl Acetate
- Raspberry Ketone (5471-51-2)
- Para-Cresyl Phenyl Acetate
- Guaiyl Acetate
- Allyl Disulfide (2179-57-9)
- Phenyl Ethyl Salicylate
- Phenylacetic Acid (103-82-2)
- Octyl Acetate (112-14-1)
- Hydroxy Citronellol (107-74-4)
- Triethyl Citrate (77-93-0)
- Diallyl Disulfide
- Isobuteric Acid
- Honey Ketone
- Turmeric Oil (8024-37-1)
- Linalyl Acetate (115-95-7)
- Tolu Balsam (9000-64-0)
- Mix Xylene
- Dementholised Oil
- Isovaleric Acid (503-74-2)
- Methyl 2-Furoate
- Iso Butyl Salicylate
- Methoxy Melonal (62439-41-2)
- Phenethyl Acetate (103-45-7)
- Prenyl Acetate
- Verdyl Propionate
- Rose Oxide (16409-43-1)
- Tea Tree Oil
- 2 Methyl 2 Methyldithio Propanal
- 1-Butanethiol (109-79-5) (C4h10s)
- Cinnamyl Isobutyrate (103-59-3)
- Aam Panna Flavour
- Butyric Acid (107-92-6)
- Camphor (76-22-2) (C10H16O)
- Caprylic Acid (C8H16O2)
- 4-Tert-Butyl Cyclohexanol (98-52-2)
- Catechol (120-80-9)
- Acetoin (513-86-0)
- Amyl Acetate
- Anisole (100-66-3)
- Acetophenone (98-86-2) (C8H8O)
- Allyl Isothiocyanate (57-06-7)
- Alpha-Copaene (3856-25-5) (C15H24)
- Benzyl Alcohol (100-51-6)
- Cycloh exanone
- 1,4- Dimethoxybenzene
- 1-Butanethiol (109-79-5) (C4H10S)
- Butyl Acetate (123-86-4)
- 2-Octanol (123-96-6) (C8H18O)
- 3-Hydroxyacetophenone (121-71-1) (C8H8O2)
- Carvacrol (499-75-2) (C10H14O)
- 4-Tert-Butylcyclohexyl Acetate (32210-23-4)
- 5-Decanolide (705-86-2) (C10H18O2)
- Acetoin (513-86-0) (C4H8O2)
- Amyl Acetate (628-63-7)
- Cis-3-Hexenyl-2-Methyl Butyrate (53398-85-9)
- Alpha-Phellandrene (99-83-2) (C10H16)
- Benzaldehyde (100-52-7) (C7H6O)
- Benzyl Benzoate (120-51-4)
- Isoamyl Acetate (123-92-2)
- Isoamyl Alcohol (123-51-3)
- Isoborneol (124-76-5) (C10H18O)
- Isobornyl Acetate (125-12-2)
- Ben-zyl Cy-anide
- Isobutyraldehyde (78-84-2) (C4H8O)
- Beta-Sitosterol (83-46-5) (C29H50O)
- Hexyl Acetate (142-92-7)
- Lemongrass Oil (8007-02-1)
- Cinnamon Oil (Liquid)
- D-Carvone (2244-16-8) (C10H14O)
- Hydroxycitronellal (107-75-5) (C10H20O2)
- Nerol Oxide (1786-08-9)
- P-Cresol (106-44-5) (C7H8O)
- Maltol (118-71-8) (C6H6O3)
- Melonal (106-72-9) (C9H16O)
- Phenethyl Propionate (122-70-3)
- Methyl 6-Methylnicotinate (5470-70-2)
- Methyl Anthranilate (134-20-3)
- Methyl Acetoacetate (105-45-3)
- Methyl Benzoate (93-58-3)
- Piperonal (120-57-0) (C8H6O3)
- Methyl Eugenol (93-15-2)
- Methyl Formate (107-31-3)
- Methyl Heptenone (110-93-0)
- Methyl Nicotinate (93-60-7)
- Methyl Phenylacetate (101-41-7)
- Propionaldehyde (123-38-6) (C3H6O)
- Alpha-Terpineol (98-55-5) (C10H18O)
- Benzylidene Acetone (122-57-6)
- Beta Pinene (18172-67-3)
- Cinnamic Acid (621-82-9)
- Coumarin (91-64-5) (C9H6O2)
- Diacetyl (0431-03-08) (C4H6O2)
- Dimethyl Sulfide (75-18-3)
- D-Limonene (5989-27-5) (C10H16)
- Helional (1205-17-0) (C11H12O3)
- Heptanoic Acid (111-14-8)
- Fruity Ketal (6290-17-1)
- 4-Methylbenzaldehyde (104-87-0) (C8H8O)
- Ethyl Acetoacetate (141-97-9)
- Galaxolide (1222-05-05) (C18H26O)
- Gamma-Decalactone (706-14-9) (C10H18O2)
- Gamma-Octalactone (104-50-7) (C8H14O2)
- Ethyl Salicylate (118-61-6)
- Ethylene Glycol Diacetate
- Evernyl (C10H12O4) (Methyl Evernate)
- Farnesol (4602-84-0) (C15H26O)
- Guaiacol (90-05-1) (C7H8O2)
- 6-Methylcoumarin (92-48-8) (C10H8O2)
- Anisole (100-66-3) (C7H8O)
- Ambrocenide (C18H30O2) (Solid)
- Indene (95-13-6) (C9H8)
- Benzyl Cyanide (140-29-4)
- Ethylene Glycol Monobutyl Ether
- Myristic Acid (544-63-8)
- N-Butyraldehyde (123-72-8) (C4H8O)
- N-Propanol (71-23-8) (C3H8O)
- Lavender Oil (8000-28-0)
- Lemon Oil (8008-56-8)
- Nerolidol (7212-44-4) (C15H26O)
- Limonene (138-86-3) (C10H16)
- Linalool (78-70-6) (C10H18O)
- Octanol (111-87-5) (C8H18O)
- Lyral (31906-04-04) (C13H22O2)
- Malic Acid (6915-15-7)
- Methyl Ethyl Ketone
- Methyl Nonyl Ketone
- Methyl Tert-Butyl Ether
- Amber Fleur (Amber Floral Accord)
- 1-Butanol (71-36-3) (C4H10O)
- 2-Butanol (78-92-2) (C4H10O)
- Butyl Butyrate (109-21-7)
- Catechol (120-80-9) (C6H6O2)
- 5-Methyl-2-Hepten-4-One (81925-81-7) (C8H14O)
- Benzophenone (119-61-9) (C13H10O)
- Nootkatone (4674-50-4) (C15H22O)
- 2-Methoxy-4-4-Methyl-1,3-Dioxolan-2-Yl Phenol (68527-74-2)
- Bisabolol (C15H26O) (Levomenol)
- Citronella Oil (8000-29-1)
- Diethyl Malonate (105-53-3)
- Dihydromyrcenol (18479-58-8) (C10H20O)
- Ethyl Isobutyrate (97-62-1)
- Ethyl Linalool (10339-55-6)
- Ethyl Nicotinate (614-18-6)
- Geraniol (106-24-1) (C10H18O)
- Hedione (24851-98-7) (C13H22O3)
- Mandarin Aldehyde (20407-84-5)
- Methyl Jasmonate (39924-52-2)
- Para-Anisyl Methyl Ketone
- Resinoid Guggul (Commiphora mukul resin)
- Skatole (83-34-1) (C9H9N)
- Salicylaldehyde (90-02-8) (C7H6O2)
- Sandalore (C14H26O) (3-Methyl-5-(2,2,3-trimethylcyclopent-3-en-1-yl)pentan-2-ol)
- Thymol (89-83-8) (C10H14O)
- Tonalide (21145-77-7) (C18H26O)
- Vanillin (121-33-5) (C8H8O3)
- Vanillylacetone (122-48-5) (C11H14O3)
- Veratrole (91-16-7) (C8H10O2)
- Diethyl L-(+)-Tartrate (87-91-2) (C8H14O6)
- Valencene (4630-07-03) (C15H24)
- 10-Undecenal (112-45-8) (C11H20O)
- 2-Chlorobenzaldehyde (89-98-5) (C7H5ClO)
- 2-Methylbutanoic Acid (116-53-0)
- 2-Phenylethanol (60-12-8) (C8H10O)
- 3-Methoxy-3-Methyl-1-Butanol (56539-66-3) (C6H14O2)
- Aldehyde C-10 (112-31-2)
- Aubepine (0123-11-5) (Para-Anisaldehyde)
- Benzyl Mercaptan (100-53-8)
- Beta-Citronellol (106-22-9) C10H20O)
- Cinnamyl Alcohol (104-54-1)
- Diethyl Succinate (123-25-1)
- Ethyl Butyrate (105-54-4)
- Furfural (98-01-1) (C5H4O2)
- Heptanal (111-71-7) (C7H14O)
- Hexanal (66-25-1)
- Indole (120-72-9)
- Ethylene Brassylate (105-95-3)
- 2-Methoxynaphthalene (93-04-9) (C11H10O)
- Benzyl Acetone (2550-26-7)
- Cedarwood Oil (8000-27-9)
- Amyl Salicylate (2050-08-0)
- Benzyl Acetate (140-11-4)
- Isobutanol (78-83-1) (C4H10O)
- 4-Methoxyphenol (150-76-5) (C7H8O2)
- delta-3-carene (13466-78-9) (C10H16)
- Acrolein (0107-02-08) (C3H4O)
- 2-Hexenal (6728-26-3) (C6H10O)
- Cyclohexanol (108-93-0) (C6H12O)
- Beta-Damascenone (23696-85-7) (C13H18O)
- Ethyl Acetate (141-78-6)
- Ethyl Acetate (141-78-6)
- Eugenol (97-53-0) (C10H12O2)
- Hexanal (66-25-1) (C6H12O)
- Indole (120-72-9) (C8H7N)
- Iso E Super
- Isoamyl Laurate (6309-51-9)
- Methyl Cinnamate (103-26-4)
- Tert-Amyl Alcohol (75-85-4)
- 1,4-Dimethoxybenzene (150-78-7) (C8H10O2)
- Trans,Trans-2,4-Hexadienal (142-83-6) (C6H8O)
- Nonanal (124-19-6) (C9H18O)
- Germacrene D (23986-74-5) (C15H24)
- Octanal (124-13-0) (C8H16O)
- Tabanon (13215-88-8) (C13H18O)
- Gamma-Hexanolactone (0695-06-07) (C6H10O2)
- Dl-Menthol (89-78-1) (C10H20O)
- Ethyl Octanoate (106-32-1)
- 2,5-Dimethylpyrazine (123-32-0) (C6H8N2)
- 2-Methyl Pyrazine (109-08-0)
- Alpha-Hexylcinnamaldehyde (101-86-0) (C15H20O)
- Pine Oil (liquid)
- 1-Nonanol (0143-08-08) (C9H20O)
- Cardamom Aldehyde (C10H18O)
- Geosmin (C12H22O) (4,8a-Dimethyloctahydro-4a(2H)-naphthalenol)
- Ambrettolide (28645-51-4) (C16H28O2)
- Cinnamyl Isobutyrate
- 2-MethylButyraldehyde (96-17-3) (C5H10O)
- Cis-3-Hexenyl Lactate (61931-81-5)
- Trans-2-Hexenyl Acetate (2497-18-9)
- Geraniol 60 (C10H18O)
- Hercolyn D (Liquid)
- Jasmolactone (32764-98-0) (C10H16O2)
- Furfurylamine (617-89-0) (C5H7NO)
- Clove oil (8000-34-8)
- Orange oil (8008-57-9)
- Aldehyde C-18 (104-61-0)
- Ethyl Hexanoate (123-66-0)
- Delta-Dodecalactone (713-95-1) (C12H22O2)
- Fructone (10-01-6413) (C8H14O4)
- Timber Propanol (70788-30-6)
- Alpha-Terpinyl Acetate (80-26-2)
- Cashmeran (33704-61-9) (C14H22O)
- Verdoracine (14374-92-6) (C13H18)
- Orange Aldehyde (Liquid)
- Methional (3268-49-3) (C4H8OS)
- Cyclotene (80-71-7) (C6H8O2)
- Citronellyl Ethoxalate (60788-25-2)
- Alpha-Humulene (6753-98-6) (C15H24)
- Cardamom Bark Oil
- Citronellal (106-23-0) (Liquid)
- L-Limonene (5989-54-8) (C10H16)
- Turpentine Oil (8006-64-2)
- 2,6-Dimethylpyrazine (108-50-9) (C6H8N2)
- Ambergris Ketone (54464-57-2)
- 4-Terpineol (562-74-3) (4-Carvomenthenol)
- Phenylacetaldehyde 85% (C8H8O)
- Methyl-3-Methylthio Propionate (13532-18-8)
- Veramoss (C10H12O4) (Solid)
- Indobranide (Solid) (354.53 g/mol)
- American Peppermint Oil
- Ethyl Oleate (111-62-6)
- Gamma-Damascone (35087-49-1) (C13H20O)
- Menthol Crystal (C10H20O)
- Globanone (C16H28O) (Liquid)
- Sandranol (C14H24O) (Liquid)
- Camphor Oil (Liquid)
- Butyl Formate (592-84-7)
- Menthol Oil (Liquid)
- Trimethyl amine 25% aa aqueous
- Isoamyl Formate (110-45-2)
- Isovaleraldehyde (590-86-3) (C5H10O)
- 1-Hexanol (111-27-3)
- 1-Pentanol (71-41-0) (C5H12O)
- 2-Acetylpyrazine (22047-25-2) (C6H6N2O)
- 2-Acetylpyridine (1122-62-9) (C7H7NO)
- 2-Acetylpyrrole (1072-83-9) (C6H6N2O)
- 2-Heptanol (543-49-7)
- 2-Pentanol (6032-29-7) (C5H12O)
- 2-Propanethiol (75-33-2)
- 2'-Aminoacetophenone (551-93-9)
- 3-Hydroxybenzoic Acid
- 3-Pentanone (96-22-0)
- 3,4-Dimethoxybenzaldehyde (120-14-9) C9H10O3
- 4-Hydroxybenzaldehyde (123-08-0) (C7H6O2)
- 4-Methoxyacetophenone (0100-06-01)
- 4-Methylacetophenone (122-00-9)
- Aldehyde C-12 Mna (110-41-8)
- Gamma-Undecalactone (104-67-6) (C11H20O2)
- Allyl Cyclohexyl Propionate
- Alpha-Amyl Cinnamaldehyde (122-40-7)
- Alpha-Bisabolol (515-69-5) (C15H26O)
- Alpha-Damascone (43052-87-5)
- Alpha-Farnesene (502-61-4)
- Alpha-Isomethyl Ionone (127-51-5)
- Anisyl Acetate (104-21-2)
- Benzyl Salicylate (118-58-1)
- Borneol (507-70-0) (C10H18O)
- Camphene (79-92-5) (C10H16)
- Citral (5392-40-5) (C10H16O)
- Cuminaldehyde (0122-03-02) (C10H12O)
- Cyclopentadecanone (502-72-7)
- Cyclopentanone (120-92-3) (C5H8O)
- Dibenzyl Ether
- Dimethyl Trisulfide (3658-80-8)
- Disodium Succinate
- Ethyl Methylphenylglycidate (77-83-8)
- Ethyl Vanillin (121-32-4)
- Furaneol (C6H8O3) (128.13 g/mol)
- Gamma-Valerolactone (108-29-2)
- Geranic Acid
- Geranyl Acetate
- Geranyl Acetate (105-87-3)
- Hexyl Salicylate (6259-76-3)
- Isobutyric Acid (79-31-2)
- Isophorone (78-59-1) (C9H14O)
- Jasmone (0488-10-8) (C11H16O)
- L-Carvone (6485-40-1) (C10H14O)
- Longifolene (475-20-7)
- M-Cresol (108-39-4) (C7H8O)
- Methoxyphenyl Methyl Butanone
- Methyl Acetate (79-20-9)
- Methyl Butyrate (623-42-7)
- Methyl Isobutyl Ketone
- Methyl Laurate (111-82-0)
- Musk Ambrette (83-66-9)
- Musk Ketone (81-14-1)
- Musk Xylene (81-15-2)
- N-Valeraldehyde (110-62-3)
- O-Cresol (95-48-7)
- Octyl Palmitate
- P-Cymene (99-87-6) (C10H14)
- Pentanoate (109-52-4) (C5H10O2)
- Phenetole (103-73-1)
- Phenyl Ethyl Methyl Ether
- Spilanthol (25394-57-4)
- Terpinolene (586-62-9) (C10H16)
- Tetramethylpyrazine (1124-11-4) (C8H12N2)
- Verdyl Acetate (5413-60-5)
- Traseolide (68140-48-7) (C18H26O)
- Phenylacetaldehyde (122-78-1) (C8H8O)
- Eucalyptol (470-82-6) (C10H18O)
- Myristicin (607-91-0)
- Ethyl Levulinate
- Geranyl Acetone
- Cis-3-Hexen-1-Ol (928-96-1) (C6H12O)
- Cis-3-Hexenyl Salicylate (65405-77-8)
- Caramel Furanone
- 2-Methoxy-4-Vinylphenol (7786-61-0)
- 3'-Methoxypropiophenone (37951-49-8)
- Safranal (116-26-7) (C10H14O)
- Ethyl Palmitate (628-97-7)
- Ginger Oil (8007-08-07)
- Isovanillin (621-59-0) (C8H8O3)
- Furfuryl Disulfide (4437-20-1)
- Dihydrocoumarin (119-84-6) (C9H8O2)
- Beta-Damascone (23726-91-2) (C13H20)
- Ethyl 2-Methylbutyrate (7452-79-1)
- Alpha-Terpinene (99-86-5) (Terpilene)
- Allyl Hexanoate (123-68-2)
- 4-Methoxybenzyl Alcohol (105-13-5)
- 3-Phenyl-1-Propanol (122-97-4)
- 2-Methylanisole (578-58-5)
- Alpha-Ionone (127-41-3) (C13H20O)
- Ethyl Propionate (105-37-3)
- Lysmeral (80-54-6) (C14H20O)
- Dihydro-Beta-Ionone (17283-81-7) (C13H22O)
- Disodium-5-Guanylate (5550-12-9)
- Delta-Damascone (57378-68-4)
- 2-Acetylthiophene (88-15-3)
- 2-Heptylcyclopentanone (0137-03-01) (C12H22O)
- Acetal (105-57-7) (C6H14O2)
- 2,4-Decadienal (25152-84-5)
- 4-Methyl-5-Thiazoleethanol (137-00-8) (C6H9NOS)
- Perillaldehyde (2111-75-3)
- Styrallyl Acetate (93-92-5)
- Sandalrome (28219-61-6) (Sandacanol)
- Trans-2-Hexen-1-Ol (928-95-0)
- Juniper Berries
- Iso-Damascone (39872-57-6)
- Isoeugenol (97-54-1) (C10H12O2)
- Isolongifolene (1135-66-6) (C15H24)
- Ethyl Cinnamate (103-36-6)
- Furfuryl Mercaptan (98-02-2)
- Civetone (542-46-1) (C17H30O)
- Benzyl Isothiocyanate
- Anethole (104-46-1) (C10H12O)
- Dihydromyrcene (2436-90-0)
- Benzyl Cinnamate (103-41-3)
- Caryophyllene Oxide (1139-30-6)
- Allyl Amyl Glycolate
- 2-Isobutylthiazole (18640-74-9) (C7H11NS)
- 4-Methoxy-2-Methyl Butane Thiol
- Ethyl Phenylacetate (101-97-3)
- Trans-Cinnamaldehyde (14371-10-9) (Cinnamic aldehyde)
- 3-Methyl-2-Buten-1-Ol (556-82-1)
- Alpha-Cedrene (469-61-4)
- 3-Methylpyridine (108-99-6) (C6H7N)
- Trans,Cis-2,6-Nonadienal (557-48-2) (C9H14O)
- Undec avertol
- Fenchyl Acetate (C12H20O2)
- Lavandulyl Acetate (25905-14-0)
- Isolongifolene Ketone
- Jasmin Lactone (34686-71-0)
- Beta-Caryophyllene (87-44-5) (C15H24)
- D-Camphor (464-49-3) (C10H16O)
- Dextro-Dihydrocarvone (7764-50-3)
- Cyclohexenone (108-94-1) (C6H10O)
- Cyclopentanol (96-41-3)
- Disodium 5-Inosinate (4691-65-0)
- Dihydrojasmone (1128-08-01)
- Cyclohexyl ethyl acetate
- Butyl Propionate (590-01-2)
- Amyris Oil (8015-65-4)
- Alpha-Thujone (546-80-5)
- 2,3-Dimethylpyrazine (5910-89-4) (C6H8N2)
- 2-Octanone (111-13-7) (C8H16O)
- Synthetic Musk (C12H15N3O6)
- Jaggery Flavor
- Benzyl Formate (104-57-4)
- Methoxyhexane Thiol
- Cedrat Oil
- Geranyllinalool (1113-21-9)
- Cedrat Oil
- 1-Octen-3-One (4312-99-6) (Amyl Vinyl Ketone)
- 1-Octen-3-yl Acetate (2442-10-6) (C10H18O2)
- Isoamyl Methyl Ketone
- Aldron (C17H28O) (248.4 g/mol)
- 2-Acetonaphthone (93-08-3) (C12H10O)
- 2-Methylbutylamine (96-15-1)
- Phenyl Ethyl Allyl Ether
- Methyl 10-Undecenoate (111-81-9)
- Vinyl Sulfurol
- Timber Touch (226 g/mol)
- Alpha-Methyl Cinnamaldehyde (101-39-3)
- Dimethyl Anthranilate (85-91-6)
- Tiglic Acid (80-59-1)
- 3,5,5-Trimethyl-1-Hexanol (3452-97-9)
- Farnesal (19317-11-4)
- Methyl levulinate
- Dodecanal (112-54-9) (C12H24O)
- Exaltolide (0106-02-05)
- Vertofix (32388-55-9) (C17H26O)
- Orcinol Dimethyl Ether
- Bornyl Acetate
- Saffron Oil (8022-19-3)
- Delta-Octalactone (698-76-0) (C8H14O2)
- Octahydrocoumarin (4430-31-3) (Bicyclononalactone)
- 3-Methoxy-3-Methyl Butanol
- Amyl Isovalerate
- Ethyl Isovalerate (108-64-5)
- Methyl Cedryl Ether
- Ethyl Isoamyl Ketone
- Methyl Heptanoate (106-73-0)
- Anthamber Top
- Exaltenone (14595-54-1)
- Glyceryl Tribenzoate
- Verdol (156.27 g/mol) (209 C)
- 2-MethoxyBenzoic Acid (579-75-9)
- Gera nodyle
- Hydroxyambran (118562-73-5) (C15H30O)
- Mugetanol (Liquid) (154.25 g/mol)
- Lemon Hexadiene (30640-46-1)
- Alpha-DihydroIonone (31499-72-6)
- Methyl Ionone Alpha Extra
- 4-tert-ButylCyclohexanone (98-53-3)
- Laevo-Camphor (464-48-2) (C10H16O)
- Ethyl Caprate (110-38-3)
- Diethyl Adipate
- Ce talox
- Trans-3-Hexenoic Acid (1577-18-0)
- 2-Isopropyl-N,2,3-Trimethylbutyramide (51115-67-4) (C10H21NO)
- 5-Methylfurfural (620-02-0) (C6H6O2)
- Theaspirane (36431-72-8)
- Aurantiol (89-43-0) (C18H27NO3)
- Beta-Santalene (511-59-1) (C15H24)
- Isophytol (505-32-8) (C20H40O)
- Cis-3-Hexenyl Isobutyrate
- Cis-3-Hexenyl Isobutyrate (41519-23-7)
- Velo utone
- Sandal Butenol
- 4-Methyl Guaiacol (93-51-6)
- Lilyflore (C12H16O) (176 g/mol)
- 2-Isopropyl-4-Methylthiazole (15679-13-7) (C7H11NS)
- Ethyl Isothiocyanate
- Hay Absolute (8031-00-3)
- 4-Methyl Thio Benzyl Cyanide
- Trans-2-Hexenoic Acid (13419-69-7)
- Para-Isopropyl Phenol (99-89-8)
- Beta-Caryophyllene Alcohol (472-97-9)
- Agarwood Oil (Agar Oil)
- Chilli Seed Oil
- Garlic Oil (8000-78-0)
- Ortho-Tolualdehyde (529-20-4) (120.15 g/mol)
- Polysantol (107898-54-4) (220 g/mol)
- Trimethylamine 25% Aqueous
- Undecanal (112-44-7) (C11H22O)
- Sodium Succinate Hexahydrate
- Hexahydrofarnesyl Acetone (502-69-2)
- Guaiacwood Oil (Liquid)
- 1-Hexanol (111-27-3) (C6H14O)
- Strawberry Liquid Flavour
- Tetrahydrolinalool (78-69-3) (C10H22O)
- Gum Benzoin (2593352)
- Linalyl Isobutyrate (78-35-3)
- 2-Nonanone (821-55-6) (C9H18O)
- Allyl Heptanoate (142-19-8)
- 5-Methyl-2-Phenyl-2-Hexenal (21834-92-4) (C13H16O)
- Alpha-Pinene (80-56-8) (Liquid)
- Linalyl Propionate (144-39-8)
- Anise Oil (8007-70-3)
- Cinnamon Bark Oil
- Watermelon Ketone
- Geranyl Caproate (2970191)
- Cinnamyl Acetate (103-54-8)
- Geranyl Formate (105-86-2)
- Trans-4-Decenal (65405-70-1) (C10H18O)
- Gamma-Methyl Decalactone (7011-83-8) (C11H20O2)
- L-Isopulegol (89-79-2) (C10H18O)
- Disodium Succinate Anhydrous
- Ethyl Decadienoate (3025-30-7)
- Geranyl Isobutyrate (2345-26-8)
- N-Propyl Propionate (106-36-5)
- Cyclohexyl Salicylate (25485-88-5)
- Phenyl Ethyl Butyrate
- Basil Oil (8015-73-4)
- 2-Methyl-3-Furanthiol (28588-74-1) (C5H6OS)
- Cyclamen Aldehyde (103-95-7)
- Lavandin Oil (Liquid)
- Mandarin Oil (Liquid)
- Ethylhexyl Triazone (88122-99-0)
- Grape Liquid Flavour
- Guava Liquid Flavour
- Mixed Fruit Powder Flavour
- Cherry Powder Flavour
- Mixed Fruit Liquid Flavour
- Honey Liquid Flavour
- Spearmint Powder Flavour
- Bitter Masker Ultraseal Flavour
- Spearmint Liquid Flavour
- Strawberry Liquid Flavour
- Badam Sweet Flavour
- Eucalyptus Oil (liquid)
- Clove Leaf Oil
- Isoborneol Flakes (Solid)
- Cardamom Oil (liquid)
- Almond Oil (liquid)
- Tagetes Oil (Tagetes Essential Oil)
- Ws-23 (Symcool Ws-23) (Powder)
- Armoise Oil (68991-20-8)
- Immortelle Absolute (977060-66-4)
- Beta-Phellandrene (C10H16) (3-Isopropyl-6-Methylenecyclohex-1-Ene)
- Costus Root Oil
- Sugandh Mantri Oil
- Sugandh Kokila Oil
- Sugandh Bala Oil
- Kapoor Kachri Oil
- 9-Decenal (13831858) (C10H18O)
- Spearmint Oil (8008-79-5)
- Methylchloroisothiazolinone (26172-55-4) (MCI)
- Dihydroambrettolide (109-29-5) (C16H30O2)
- Estragole (140-67-0) (C10H12O)
- Jasmine Grandiflorum (Jasmine absolute)
- Grapefruit Essential Oil
- Sandalwood (liquid) (276C)
- O-Tert-ButylCyclohexyl Acetate (88-41-5)
- Beta-Bisabolene (495-61-4) (C15H24)
- 3-Octyl Acetate (4864-61-3)
- Lilybelle (C10H16) ((S)-Limonene)
- Laevo-Linalool (126-91-0) (C10H18O)
- Ambercore (C12H24O2) (Amber Core)
- Citronellyl Phenylacetate (139-70-8)
- Acetyl Longifolene (3338-56-5)
- Lavender Fragrance Oil
- Dynascone (C13H20O) (Galbascone )
- Habanolide (C16H28O) (Musk Ketone Givaudan)
- Bourgeonal (18127-01-0) (C13H18O)
- Globalide (C16H28O2) (9-Cycloheptadecen-1-one)
- Cinnamyl Formate (104-65-4)
- L-Menthyl Lactate (61597-98-6)
- Gardamide (84434-18-4) (C12H17NO)
- 4-EthylPhenol (123-07-9) (C8H10O)
- Styrax Essential Oil
- Verbenone (80-57-9) (C10H14O)
- Isomuscone (C16H30O) (Cyclohexadecanone)
- 10-Undecen-1-Ol (112-43-6) (C11H22O)
- Herbac (25304-14-7) (C10H18O)
- Gum Turpentine Oil
- Linalyl Formate (115-99-1)
- Ginger Oleoresin (C17H26O4)
- Sandal Butanol (liquid)
- Ketone 101 (liquid)
- Methyl Atrarate (4707-47-5)
- Beta Terpineol (138-87-4)
- Sandal Cyclohexanol (66068-84-6)
- 2-Pentylfuran (3777-69-3) (C9H14O)
- Sakura Salicylate (Liquid)
- 2-Ethyl Fenchol (18368-91-7)
- Musk T (C15H26O4)
- Rhovanil (C8H8O3) (Vanillin)
- L-Citronellol (7540-51-4) (C10H20O)
- Nopyl Acetate (35836-72-7)
- Phenyl Propyl Cinnamate
- Phenyl Propyl Acetate
- Cardamom Essential Oil
- Phenyl Ethyl Isoamyl Ether
- Pentyl Benzoate (2049-96-9)
- Allyl Octanoate (4230-97-1)
- Camphor Essential Oil
- Vetiver Essential Oil
- Wormwood Essential Oil
- 2-Methyltetrahydrofuran-3-One (3188-00-9) (liquid)
- Irisnitrile (C9H15N) (Non-2-enenitrile)
- L-Rose Oxide (3033-23-6)
- Benzyl Isovalerate (103-38-8)
- Ethyl Heptoate (C9H18O2)
- Cinnamyl Butyrate (103-61-7)
- Cinnamyl Isovalerate (140-27-2)
- Ambrinol S (C16H28O)
- Cinnamyl Propionate (103-56-0)
- Mandaril (C13H21N) (Citrus reticulata oil)
- Jacinthaflor (C10H12O2) (2-Methyl-4-phenyl-1,3-dioxolane)
- Nerolione (C11H10O2) (1-(3-Methyl-benzofuran-2-yl)-ethanone)
- Citronellyl Isovalerate (68922-10-1)
- Sultanene (C11H12O2) (Ethyl 2-Cyclopentene-1-Acetate)
- Corps Racine (C14H15N)
- Brahmanol (C13H24O ) (B,2,2,3-Tetramethylcyclopent-3-ene-1-butanol)
- Heptyl Butyrate (5870-93-9)
- Mintonat (C10H16O2) (Mint formate ester)
- Isoamyl Caproate (2198-61-0)
- Isobutyl Isovalerate (589-59-3)
- WS-3 (C13H25NO) (Liquid)
- 2-Methyl-4-Propyl-1,3-Oxathiane (C8H16OS) (Tropathiane)
- Menthofuran (494-90-6) (C10H14O)
- Thymol Crystals (C10H14O)
- N-Amyl Butyrate (540-18-1)
- N-Amyl Isobutyrate (2445-72-9)
- Amber Xtreme (C18H32O)
- Octyl Butyrate (110-39-4)
- Bisabolene (495-62-5) (C15H24)
- Lime Oxide (73018-51-6)
- Padma (101-48-4) (Phenyl Acetaldehyde Dimethyl Acetal_
- Para Anisyl Propionate
- Ambrofix (C16H28O) (Ambroxide)
- Acetal CD (C11H22O2)
- Rosaphen (C12H18O) (Liquid)
- Bacdanol (C14H24O) (Liquid)
- 2,3,5-Trimethylpyrazine (14667-55-1) (C7H10N2)
- Gamma-Terpineol (586-81-2) (C10H18O)
- Garlic Essential Oil
- Benzoin Sumatra Resinoid
- Geranyl Isovalerate (109-20-6)
- Raspberry Extract (C14H6O8)
- Heptyl Acetate (0112-06-01)
- Cinnamon Powder (C9h6O)
- Octyl Isobutyrate (109-15-9)
- Geranyl Formate 95%
- Treemoss Absolute (Solid)
- Geranyl Propionate 98%
- Ambrette Seed Absolute
- Cinnamyl Cinnamate (122-69-0)
- Cis-3-Hexenyl Tiglate (67883-79-8)
- Apple Green Flavour
- Phenylhexanol (55066-48-3) (C12H18O)
- Coriander Seed Essential Oil
- Apricot Liquid Flavour
- Cypriol Essential Oil
- Bubble Gum Powder Flavour
- Geraniol Extra (C10H18O)
- Cherry Liquid Flavour
- Cis-3-Hexenyl Isovalerate (35154-45-1)
- Date Liquid Flavour
- Fig Liquid Flavour
- Grape Powder Flavour
- Kiwi Liquid Flavour
- Ambertal K (Solid)
- Lychee Powder Flavour
- Papaya Liquid Flavour
- Passion Fruit Liquid Flavour
- Pineapple Fresh Liquid Flavour
- Astromeran (C14H20O) (Cashmeran)
- Astrone (C12H22O) (Koavone)
- Camphor GF (C10H16O)
- Captaite (Fruitate) (Fruity Carboxylate)
- Chandanol (C15H24O) (Sandalwood alcohol)
- Frescamenth (C10H18O) (Isobutylmenthone)
- Limeox (C10H16O) (2,6-Dimethyl-2,6-octadien-8-one)
- Limofine STW (C10H16O)
- Mysornol (C13H22O) (Mysornol AB)
- Nectanone (C11H16O) (Nectanone)
- Nerolin Bromelia (C12H12O)
- Sandalica (C16H28O) (Isobornyl cyclohexanol)
- Pomegranate Liquid Flavour
- Plum Liquid Flavour
- Sealone (Solid) (Marine aldehyde)
- Shaktinol (Liquid) (Synthetic sandalwood alcohol)
- Mojito Flavour (Mojito Aroma)
- Shivanol (C16H28O) (Synthetic sandalwood alcohol)
- Isoborneol Powder (154.25 g/mol)
- Rose Nitrile (Liquid)
- Almond Powder Flavour
- Cashew Fruit Liquid Flavour
- Honey Dew Flavour
- Coconut Powder Flavour
- Yogurt Flavour (Yogurt Aroma)
- Coconut Liquid Flavour
- Cookies Flavour (Cookies Aroma)
- Hazelnut Liquid Flavour
- Peanut Liquid Flavour
- Pistachio Powder Flavour
- Fenugreek Oil (Trigonella foenum-graecum Oil)
- Aam Panna Flavour
- Pistachio Liquid Flavour
- Blue Raspberry Flavour
- Walnut Liquid Flavour
- Chaat Cola Flavour
- Vanilla Caramel Flavour
- Aniseed Liquid Flavour
- Cardamom Oleoresin Flavour
- Aniseed Powder Flavour
- Biryani Liquid Flavour
- Black Pepper Liquid Flavour
- Chicken Liquid Flavour
- Clove Flavour (Clove oil component)
- Cinnamon Liquid Flavour
- Cinnamon Powder Flavour
- Fennel Flavour (Fennel Essence)
- Ginger Liquid Flavour
- Cinnamon Oleoresin Flavour
- Coriander Oleoresins Flavour
- Cumin Oleoresin Flavour
- Fennel Oleoresin Flavour
- Nutmeg Oleoresin Flavour
- Rosemary Oleoresin Flavour
- Jeera Flavour (Jeera essence)
- Paan Bahar Flavour
- Rabdi Flavour (Sweet rabdi flavour)
- Lemon Juice Flavour
- Rum Flavour (Rum Aroma)
- Custard Liquid Flavour
- Whisky Flavour (Powder)
- Khus Flavour (Khus Essence)
- Kulfi Liquid Flavour
- Paan Masala Flavour
- Jasmine Essential Oil
- Coriander Liquid Flavour
- Garlic Liquid Flavour
- Tomato Liquid Flavour
- Peppermint Liquid Flavour
- Cocoa Powder Flavour
- Cocoa Liquid Flavour
- Peppermint Powder Flavour
- Coffee Powder Flavour
- Coffee Liquid Flavour
- Lemon Liquid Flavour
- Honey Powder Flavour
- Malt Flavour (Malt Aroma)
- Lemon Powder Flavour
- Caramel Powder Flavour
- Caramel Liquid Flavour
- Mango Powder Flavour
- Mango Liquid Flavour
- Pineapple Liquid Flavour
- Pineapple Powder Flavour
- Raspberry Liquid Flavour
- Lime Liquid Flavour
- Lime Powder Flavour
- Milk Flavour (Milk Aroma)
- Butter Powder Flavour
- Butter Liquid Flavour
- Cheese Powder Flavour
- Apple Liquid Flavour
- Apple Powder Flavour
- Liquorice Flavour (Licorice Flavour)
- Raspberry Powder Flavour
- Orange Liquid Flavour
- Orange Powder Flavour
- Cream Flavour (Dairy Flavour)
- Vanilla Liquid Flavour
- Vanilla Powder Flavour
- Tridec-2-Enenitrile (22629-49-8) (C13H23N)
- Strawberry Powder Flavour
- Banana Liquid Flavour
- 4-4-Methoxyphenyl -2-Butanone (104-20-1) (C11H14O2)
- 4-Methoxytoluene (104-93-8) (C8H10O)
- Banana Powder Flavour
- Saffron Powder Flavour
- Dimethyl Octanyl Acetate
- Isobutyl Butyrate (539-90-2)
- Jasmine Sambac (Liquid)
- Dihydro Iso Jasmonate
- 2-Methylthiazole (3581-87-1) (Liquid)
- Benzyl Ethyl Ether
- Furfuryl Acetate (623-17-6)
- Star Anise Oil
- Lavender Fragrance (Liquid)
- Benzoin Siam Resinoid
- Helvetolide (141773-73-1) (Solid)
- Vetiver Oil Terpenes
- 1,4-Cineole (470-67-7) (Liquid)
- Cetalox (C16H28O) (solid)
- Dipropylene Glycol (25265-71-8)
- 2-Ethoxyethylamine (110-76-9) (C4H11NO)
- Paprika Oleoresin (68917-78-2)
- Industrial Chemicals
- Calcium Carbonate (471-34-1)
- Chlorinated Paraffin Wax
- Calcium Chloride
- Citric Acid Monohydrate
- Ammonium Stearate
- Ammonium Polyacrylate
- Calcium Silicate
- Copper Carbonate (Powder)
- Boron Nitride (10043-11-5)
- Calcium Oxide (1305-78-8)
- Calcium Sulfate
- Alcohol Ethoxylate
- Calcium Stearate
- Cyanoacetic Acid (372-09-8)
- Ammonium Bifluoride
- Sodium Ethoxide (141-52-6)
- Caustic Soda Flakes
- Calcium Chloride Dihydrate
- Ammonium Sulfamate
- Capric Acid
- Cobalt Sulphate
- Sodium Chloride (7647-14-5)
- Sodium Stannate
- Sodium Hydroxide
- Sodium Polyacrylate
- Sodium Metabisulfite
- Propionic Acid (79-09-4)
- Sodium Gluconate (527-07-1)
- Sodium Silicate (1344-09-8)
- Zirconium Oxide
- Melamine Cyanurate
- Sodium Methoxide (124-41-4)
- Sorbitan Trioleate
- Non Ferric Alum
- Magnesium Sulphate
- Sodium Hydrosulfide
- Glutaric Acid (110-94-1)
- Sodium Cryolite (15096-52-3)
- Magnesium Nitrate
- Lithium Carbonate (554-13-2)
- Zinc Hydroxide
- Magnesium Fluoride
- Sodium Thiosulphate (Na2O3S2)
- Sodium Lauryl Sulfate
- Lactic Acid
- Sorbitan Monooleate
- Hydrochloride Chemical
- Mono Sodium Phosphate
- Hydrofluoric Acid (7664-39-3)
- Methyl Ethyl Ketoxime
- Hydrazine Hydrate (10217-52-4)
- Magnesium Carbonate (546-93-0)
- Neopentyl Glycol
- Magnesium Oxide (1309-48-4)
- Hydrogen Peroxide
- Methylene Chloride
- Phosphorus Pentoxide (1314-56-3)
- Methylamine Hydrochloride (593-51-1)
- Magnesium Silicate (1343-88-0)
- Boric Acid (10043-35-3)
- Hydrochloric Acid (7647-01-0)
- Tricalcium Phosphate
- Zinc Stearate
- Fumaric Acid (110-17-8)
- Zinc Carbonate (5263-02-05)
- Titanium Dioxide
- Triphenyl Phosphite
- Silicone Emulsion
- Isopropyl Alcohol (67-63-0)
- Boron Carbide (12069-32-8)
- Silica Gel (SiO2)
- Itaconic Acid
- Xanthan Gum
- Zinc Phosphate (7779-90-0)
- Sodium Formate (141-53-7)
- Methyl Acrylate
- Copper Nitrate (3251-23-8)
- Silicon Dioxide (7631-86-9)
- Sodium Nitrate
- Triphenyl Phosphate
- Hydrofluoric Acid 20%
- Hydrofluoric Acid 40%
- Aluminium Fluoride Anhydrous
- Sodium Hexafluoroaluminate
- Aluminium Fluoride Trihydrate
- Ammonium Fluoride
- Potassium Aluminium Fluoride
- Sodium Bifluoride (1333-83-1)
- Barium Carbonate (513-77-9)
- Barium Hydroxide Anhydrous
- Barium Hydroxide Octahydrate
- Barium Hydroxide Monohydrate
- Sodium Hydrosulfide Hydrate
- Sodium Hydrosulfide Anhydrous
- Phenylhydrazine Hydrochloride (59-88-1)
- 5-Nitroisophthalic Acid
- Acetamide (60-35-5) (C2H5NO)
- 3-Nitrobenzaldehyde (99-61-6)
- Potassium Hexafluoroaluminate
- Potassium Fluoride Anhydrous
- Ammonium Tetrafluoroborate
- Fluoroboric Acid
- Ammonium Fluorosilicate
- Hexafluorosilicic Acid
- Barium Nitrate (10022-31-8)
- Magnesium Fluoride (7783-40-6)
- Tin Fluoride
- Sodium Tetrafluoroborate (13755-29-8)
- Ammonium Bifluoride (1341-49-7)
- Barium Peroxide (1304-29-6)
- Phenylhydrazine (100-63-0) (C6H8N2)
- Phthalimide (85-41-6) (C8H5NO2)
- Benzoguanamine (91-76-9)
- Pyridinium Chlorochromate (26299-14-9)
- Pyridine Sulfur Trioxide
- Mal-eic Hydr-azide
- 2-Bromoethylamine Hydrobromide
- 4-Phenylbutyric acid
- 3-Indoleacetic acid
- 4-Dimethylaminobenzaldehyde (24057-28-1)
- Pyridinium P-Toluenesulfonate (24057-28-1) (C12H13NO3S)
- Potassium Bifluoride
- Potassium Tetrafluoroborate
- Barium Sulphide
- 3-Nitrophthalic Acid (0603-11-2)
- 4-Nitrophthalic Acid (610-27-5)
- Mucic Acid (526-99-8)
- 4-Chlorophenylhydrazine Hydrochloride
- Potassium Phthalimide
- N-2-Hydroxyethyl Phthalimide
- N-Hydroxymethyl Phthalimide
- Homophthalic acid
- Homophthalic Anhydride
- Dl-Camphorquinone (10373-78-1)
- 2-Benzylacrylic Acid
- Triphenylphosphine Oxide
- Dibenzyl Phosphate
- Cobalt Chloride Anhydrous
- Cobalt Chloride Hexahydrate
- Cobalt Nitrate (10141-05-06)
- Cobalt Sulfate Anhydrous
- Cobalt Sulphate Heptahydrate
- Zirconium Octoate (C32H64O8Zr)
- Zirconium Sulphate Anhydrous
- Biphenyl (92-52-4) (C12H10)
- 1,4-Naphthoquinone (130-15-4) (C10H6O2)
- Borax Pentahydrate (11130-12-4)
- 2-Mercaptopyridine N-Oxide Sodium Salt
- 2-Methyl-4-Nitroaniline (99-52-5) (C7H8N2O2)
- Calcium Carbide (75-20-7)
- Calcium Chloride Anhydrous
- Calcium Silicide (12013-56-8)
- Calcium Tungstate (7790-75-2)
- 2-Ethylanthraquinone (84-51-5) (C16H12O2)
- 2-Methylimidazole (693-98-1) (C4H6N2)
- 3-Nitrobenzoic Acid (121-92-6)
- 4-Chlorophenol (106-48-9) (C6H5ClO)
- 4-Hydroxybenzophenone (1137-42-4) (C13H10O2)
- 4-Nitroaniline (0100-01-06) (C6H6N2O2)
- Ammonium Benzoate (1863-63-4)
- Ammonium Sulphite (10196-04-0)
- Ammonium Thiocyanate (1762-95-4)
- Acetoacetanilide (0102-01-02) (C10H11NO2)
- Ammonium Thiosulphate (7783-18-8)
- Anthraquinone (84-65-1) (C14H8O2)
- Barium Chloride (10361-37-2)
- Barium chromate (10294-40-3)
- Aluminum Acetylacetonate (13963-57-0)
- Aluminum Nitride (24304-00-5)
- Ammonium Molybdate (H8MoN2O4)
- Benzyltriethylammonium Chloride (56-37-1)
- Cryptohalite (16919-19-0) (Ammonium Fluorosilicate)
- Hexachloroethane (67-72-1) (C2Cl6)
- Lead Dioxide (1309-60-0)
- Lead Nitrate (10099-74-8)
- Lead Zirconium Titanate
- Nickel Acetate Anhydrous
- Nickel Chloride (7718-54-9)
- Nickel Sulfate Anhydrous
- Limestone (1317-65-3) (100.09 g/mol)
- Nitrilotriacetic Acid (139-13-9)
- Lithium Acetate Dihydrate
- Lithium Chloride Anhydrous
- Lithium Fluoride Anhydrous
- Lithium Hydroxide Anhydrous
- Lithium Iodide Anhydrous
- O-Aminophenol (95-55-6) (C6H7NO)
- O-Phenylenediamine (95-54-5) (C6H8N2)
- Lithium Nitrate Anhydrous
- O-Toluic Acid (118-90-1)
- Magnesium Chloride Anhydrous
- Oxalic Acid Anhydrous
- Para-Toluenesulphonic Acid Anhydrous
- Manganese Chloride Anhydrous
- Mercuric Chloride (7487-94-7)
- Mercuric Nitrate Monohydrate
- Picric Acid (88-89-1)
- Potassium Permanganate (7722-64-7)
- Mica (12001-26-2) (398.31 g/mol)
- 3-Nitrochlorobenzene (121-73-3) (C6H4ClNO2)
- Pumice (1332-09-08) (60.08 g/mol)
- Pyridinium Dichromate (20039-37-6)
- Aluminium Nitrate (213.01 g/mol)
- Ammonium Bisulfite (10192-30-0)
- Chromic Acid (7738-94-5)
- Borax Anhydrous (1330-43-4)
- Copper Acetate (142-71-2)
- Copper Chloride (134.45 g/mol)
- Copper Naphthenate (C21H36CuO4)
- Copper Nitrate Trihydrate
- Cupric Chloride (7447-39-4)
- Chloroacetic Acid (79-11-8)
- Denatonium Benzoate (3734-33-6)
- Dimethylglyoxime (95-45-4) (C4H8N2O2)
- Dimethylurea (88.11 g/mol) (202 Degree C)
- Diphenyl Diselenide (1666-13-3)
- Dolomite (16389-88-1) (184.41 g/mol)
- Ethylenediaminetetraacetic Acid (60-00-4)
- Graphite (7782-42-5) (C)
- Guanidine Nitrate (506-93-4)
- Ferric Alum Dodecahydrate
- Ferric Nitrate (7782-61-8)
- Hydrazine Sulphate (10034-93-2)
- Fluorspar (7789-75-5) (CaF2)
- Potassium Acetate (0127-08-02)
- Potassium Aluminium Sulphate
- Potassium Chromate (7789-00-6)
- Potassium Ferrocyanide Anhydrous
- Potassium Fluoride Anhydrous
- P-Toluenesulfonic acid (C7H8O3S)
- Sodium Amide (7782-92-5)
- Sebacic Acid (111-20-6)
- Sodium Azide (26628-22-8)
- Sodium Bisulphate Anhydrous
- Sodium Bromide Anhydrous
- Sodium Chromate (7775-11-3)
- Sodium Tert-Pentoxide (14593-46-5)
- Sodium Hydride (7646-69-7)
- Sodium Thiosulphate Anhydrous
- Quadrol (102-60-3) (Entprol)
- Sodium Cyanate (917-61-3)
- Tetrabutylammonium Fluoride Anhydrous
- Tetrabutylammonium Fluoride Trihydrate
- Strontium Chloride Anhydrous
- Strontium Fluoride (7783-48-4)
- Strontium Nitrate (10042-76-9)
- Succinic Acid (110-15-6)
- Succinic Anhydride (108-30-5)
- Tolyltriazole (29385-43-1) (C7H7N3)
- Tosyl Chloride (98-59-9)
- Trichloroacetic Acid (76-03-9)
- Vanadyl Sulfate Anhydrous
- Triphenylphosphine (603-35-0) (C18H15P)
- Zinc Chloride Anhydrous
- 2-Chlorobenzoic Acid (118-91-2)
- 2-Cyanoacetamide (107-91-5) (C3H4N2O)
- 2-Naphthol (135-19-3) (C10H8O)
- 2,2-Bis Hydroxymethy Propionic Acid
- 3,5-Dinitrobenzoic Acid (99-34-3)
- Diphenyl Amine (122-39-4)
- Ferrocene (102-54-5) (C10H10Fe)
- Iodobenzene Diacetate (3240-34-4)
- Levulinic Acid (123-76-2)
- Mercury Sulfide (1344-48-5)
- Octadecane (593-45-3) (C18H38)
- Stannous Sulfate (7488-55-3)
- Calcium Naphthenate (61789-36-4)
- 2,6-Dichloroaniline (608-31-1) (C6H5Cl2N)
- Sodium 2-Ethylhexanoate (19766-89-3)
- Dimethyl Aminoterephthalate (5372-81-6)
- 4-Methylbenzoic Acid (99-94-5)
- Vanadium Trioxide (1314-34-7)
- Sodium Vinylsulfonate (3039-83-6)
- Magnesium Chloride Hexahydrate
- Iodic Acid (7782-68-5)
- Strontium Chloride Hexahydrate
- Sodium Dichromate Dihydrate
- 4-Isopropylcatechol (2138-43-4) (C9H12O2)
- Tetramethylammonium Hydroxide (75-59-2)
- Meta Nitro Para Toluidine
- Lead Acetate Trihydrate
- 4-Chloro-2-Nitroaniline (C6H5ClN2O2) (172.57 g/mol)
- Lithium Bromide Anhydrous
- Neodecanoic Acid (26896-20-8)
- Potassium Hydrogen Oxalate
- Sodium DiethylDithioCarbamate (148-18-5)
- Sodium Hydroxide Pellets AR
- 2-Amino-4-Chlorodiphenyl Ether (93-67-4)
- Oxalic Acid Dihydrate
- Sodium Dichromate Anhydrous
- Para-Toluenesulphonic Acid Monohydrate
- 1,3-Benzenedisulfonic Acid (98-48-6)
- 3-Hydroxy-N-Phenyl-2-Naphthamide (92-77-3) (C17H13NO2)
- Leucoquinizarin (476-60-8) (C14H10O4)
- Magnesium Ingots (24.305 g/mol)
- Ferric Alum (FeH28NO20S2)
- Alkyl Benzene Sulfonate
- Palmitoyl Chloride (112-67-4)
- Cobalt Naphthenate (61789-51-3)
- 1,2-Diaminopropane (78-90-0) (C3H10N2)
- 1,4-Butynediol (110-65-6) (C4H6O2)
- Bis(2-Ethylhexyl) Hydrogen Phosphate
- 2-Chloroethanol (0107-07-03) (C2H5ClO)
- 2-Ethylaniline (578-54-1) (C8H11N)
- Bromoform (75-25-2) (CHBr3)
- 2-Ethylhexyl Nitrate (27247-96-7)
- 2-Mercaptoethanol (60-24-2) (C2H6OS)
- 2-Methoxyethanol (109-86-4) (C3H8O2)
- 2,4,6-Trimethylpyridine (108-75-8) (C8H11N)
- 3-2-Aminoethylamino Propyl Trimethoxysilane
- 4-Heptanone (123-19-3) (C7H14O)
- 4-Methylmorpholine N-Oxide (7529-22-8)
- 4-Nitrotoluene-2-Sulfonic Acid (0121-03-09)
- Carbon Disulfide (75-15-0)
- Acetic Acid (64-19-7)
- Acetonitrile (75-05-8) (C2H3N)
- EDTA Tetrasodium Salt Anhydrous
- EDTA Tetrasodium Salt Anhydrous
- Ethyltriphenylphosphonium Iodide (4736-60-1)
- Hydrocarbon Resin (500 g/mol)
- Butyltriphenylphosphonium Bromide (1779-51-7)
- Potassium Silicofluoride (16871-90-2)
- Manganese Monoxide (1344-43-0)
- 1,3-Dihydro-2h-Benzimidazole-2-Thione (583-39-1) (C7H6N2S)
- Sodium Isopropoxide (683-60-3)
- Silicotungstic Acid (11130-20-4)
- Ammonium Heptamolybdate (12027-67-7)
- Acetyl Chloride (75-36-5)
- Acetylacetone (123-54-6) (C5H8O2)
- Allyl Alcohol (107-18-6)
- Allyl Chloride (0107-05-01)
- Benzotrichloride (98-07-7) (C7H5Cl3)
- Benzyl Chloride (100-44-7)
- Gamma-Butyrolactone (96-48-0) (C4H6O2)
- Hexamethyldisilazane (999-97-3) (C6H19NSi2)
- Morpholine (110-91-8) (C4H9NO)
- N-Butyl Chloride (109-69-3)
- N-Butylamine (109-73-9) (C4H11N)
- N-Butyllithium (109-72-8) (C4H9Li)
- N-Hexane (110-54-3) (C6H14)
- N-Pentane (109-66-0) (C5H12)
- Nitrobenzene (98-95-3) (C6H5NO2)
- Nonylphenol (84852-15-3) (C15H24O)
- Octylamine (111-86-4) (C8H19N )
- M-Toluidine (108-44-1) (C7H9N)
- Oleum (8014-95-7) (H2SO4.SO3)
- Hexafluorophosphoric Acid (16940-81-1)
- Petroleum Ether (8032-32-4)
- Mesitylene (108-67-8) (C9H12)
- Methanesulfonic Acid (75-75-2)
- Phosphorus Oxychloride (10025-87-3)
- Phosphorus Trichloride (7719-12-2)
- Methyl Iodide (74-88-4)
- Methyl Isonicotinate (2459-09-8)
- Methyl Isopropyl Ketone
- Methyl Ricinoleate (141-24-2)
- 1 2-Dichloroethane (0107-06-02)
- 18-Crown-6 (17455-13-9) (C12H24O6)
- Chloromethyl Methyl Ether
- Chlorosulfonic Acid (7790-94-5)
- Boron Trifluoride Etherate
- Cis-2-Butene-1,4-Diol (6117-80-2) (C4H8O2)
- Calcium Petroleum Sulfonate
- Cresylic Acid (1319-77-3)
- Cumene Hydroperoxide (80-15-9)
- Acetyl Bromide (506-96-7)
- Monoethylamine (75-04-7) (C2H5NH2)
- N-N-N-N-Tetrakis 2-Hydroxypropyl Ethylenediamine
- Naphthenic Acid (1338-24-5)
- Oleylamine (112-90-3) (C18H37N)
- Alkyl Phenol Ethoxylate
- Chlorine Dioxide (10049-04-04)
- Diethanolamine (111-42-2) (C4H11NO2)
- Diethyl Carbonate (105-58-8)
- Diiodomethane (75-11-6) (CH2I2)
- Diacetone Alcohol
- Diisopropanolamine (110-97-4) (C6H15NO2)
- Dimethyl Amine (124-40-3)
- Dibromomethane (74-95-3) (CH2Br2)
- Dimethyl Oxalate (553-90-2)
- Dimethylformamide (68-12-2) (C3H7NO)
- Diphenyl Oxide (101-84-8)
- Dodecane (112-40-3) (C12H26)
- Ethylcyclohexane (1678-91-7) (C8H16)
- Diethyl Dipropylmalonate (6065-63-0)
- Diethylaminoethanol (100-37-8) (C6H15NO)
- Diacetone Alcohol (123-42-2)
- Fluoroboric Acid (16872-11-0)
- Formic Acid (64-18-6)
- Polyol (62.07 g/mol) (197.3 Degree C)
- Polyphosphoric Acid (8017-16-1)
- Octyltriethoxysilane (2943-75-1) (C14H32O3Si)
- Pyrrolidine (123-75-1) (C4H9N)
- Pine Tar (8011-48-1)
- Sodium Isopropylxanthate (140-93-2)
- Stearoyl Chloride (112-76-5)
- Thionyl Chloride (7719-09-07)
- Titanium Tetrachloride (7550-45-0)
- Sulfolane (126-33-0) (C4H8O2S)
- Benzisothiazolinone (2634-33-5) (C7H5NOS)
- Sulphuryl Chloride (7791-25-5)
- Tributyl Phosphate (126-73-8)
- Tert-Butyl Alcohol (75-65-0)
- Tert-Butyl Hydroperoxide (75-91-2)
- Tert-Butylamine (75-64-9) (C4H11N)
- Tert-Butylbenzene (98-06-6) (C10H14)
- Triethyl Phosphite (122-52-1)
- Triethylene Glycol (112-27-6)
- Triethylene Glycol Monobutyl Ether
- Triethylene Glycol Monomethyl Ether
- Triethylsilane (617-86-7) (C6H16Si)
- Triisopropanolamine (122-20-3) (C9H21NO3)
- Trimethyl Borate (121-43-7)
- Trixylenyl Phosphate (25155-23-1)
- 1-Methylimidazole (616-47-7) (C4H6N2)
- 1,2-Dichloropropane (78-87-5) (C3H6Cl2)
- 1,8-Diazabicyclo-5.4.0-7-Undecene (6674-22-2) (C9H16N2)
- 2-(2-Aminoethoxy)Ethanol (0929-06-06) (C4H11NO2)
- 2-Amino-2-Methyl-1-Propanol (124-68-5) (C4H11NO)
- 2-Ethylhexylamine (104-75-6) (C8H19N)
- 2-Fluoroaniline (348-54-9) (C6H6FN)
- 2-Methyltetrahydrofuran (96-47-9) (C5H10O)
- 3-Mercaptopropionic Acid (107-96-0)
- 3-Methylbenzyl Chloride (620-19-9)
- Chlorotrimethylsilane (75-77-4) (C3H9ClSi)
- Cyclohexyl Isocyanate (3173-53-3)
- Dibutyl Ether (142-96-1)
- Diethylene Glycol Monobutyl Ether
- Diisobutyl Ketone (108-83-8)
- Trichloroisocyanuric Acid (87-90-1)
- Ethylene Carbonate (96-49-1)
- Hydroiodic Acid (10034-85-2)
- Hypophosphorous Acid (6303-21-5)
- Isodecanol (25339-17-7) (C10H22O)
- M-Xylene (108-38-3) (C8H10)
- Methyl Oleate (112-62-9)
- N-Benzylmethylamine (103-67-3) (C8H11N)
- N-Methylaniline (100-61-8) (C7H9N)
- Triisopropyl Phosphite (116-17-6)
- Trimethyl Orthoformate (149-73-5)
- Trimethoxyphenylsilane (2996-92-1) (C9H14O3Si)
- Ethyl Magnesium Bromide
- N,N,N,N-Tetramethyl-1,3-Propanediamine (110-95-2) (C7H18N2)
- Methanesulfonyl Chloride (124-63-0)
- Dioxolane (646-06-0) (C3H6O2)
- O-Xylene (95-47-6) (C8H10)
- Crotonaldehyde (4170-30-3) (C4H6O)
- 2-Butoxyethyl Acetate (0112-07-02)
- Linear alkyl benzene
- 1,6-Dichlorohexane (2163-00-0) (C6H12Cl2)
- Methyl Soyate (67784-80-9)
- Sec-Butylbenzene (135-98-8) (C10H14)
- Cobalt Carbonate (513-79-1)
- Zinc Sulphide (1314-98-3)
- Zirconium Oxychloride (7699-43-6)
- Bismuth Trioxide (1304-76-3)
- 2-Amino-4-Chlorophenol (95-85-2) (C6H6ClNO)
- Borax Decahydrate (1303-96-4)
- Cadmium Oxide (1306-19-0)
- Boron (7440-42-8) (B)
- Calcium Bicarbonate (3983-19-5)
- Calcium Hydroxide (1305-62-0)
- Calcium Silicate (1344-95-2)
- 4-Dimethylaminopyridine (1122-58-3) (C7H10N2)
- Ammonium Tetrafluoroborate (13826-83-0)
- Aluminium Isopropoxide (555-31-7)
- Hydroxylamine Hydrochloride (5470-11-1)
- Monocalcium Phosphate Anhydrous
- N-Bromosuccinimide (0128-08-05) (C4H4BrNO2)
- N-Chlorosuccinimide (0128-09-06) (C4H4ClNO2)
- Activated Alumina (101.96 g/mol)
- Alumina Hydrate (78.00 g/mol)
- Alumina Trihydrate (156.01 g/mol)
- Aluminium Hydroxide Trihydrate
- Aluminium Silicate (Al2O5Si)
- Chromium Sulphate (392.18 g/mol)
- Colemanite (411.10 g/mol) (840 Degree C)
- Cerium Oxide (172.12 g/mol)
- Chloramine-T (205.67 g/mol) (198 Degree C)
- Cyclopentylmagnesium Bromide (33240-34-5)
- French Chalk (379.26 g/mol)
- Potassium Cryolite (258.27 g/mol)
- Potassium Titanium Oxalate
- Lead Acetate Anhydrous
- Nickel Hydroxide (12054-48-7)
- Lithium Molybdate (13568-40-6)
- Lithium Silicate (10102-24-6)
- Lithium Tetraborate Anhydrous
- Para Hydroxy Benzoic Acid
- Perlite (93763-70-3)
- Polyacrylamide (9003-05-08) ((C3H5NO)n)
- Potassium Oxalate (583-52-8)
- Potassium Persulfate (7727-21-1)
- Potassium Tetraborate Tetrahydrate
- Lead Carbonate (598-63-0)
- Mercuric Oxide Red
- Barium Titanate (12047-27-7)
- Bromocresol purple (115-40-2)
- 2-Methoxy-5-Nitroaniline (99-59-2) (C7H8N2O3)
- Barium Azide (18810-58-7)
- Ammonium Citrate Dibasic
- Potassium Thiocyanate (333-20-0)
- Potassium Titanate (12030-97-6)
- Molybdenum Disulfide (1317-33-5)
- Molybdenum Trioxide (1313-27-5)
- Chromium Chloride (39345-92-1)
- Chromium Nitrate (13548-38-4)
- Chromium Oxide (11118-57-3)
- Boron Oxide (1303-86-2)
- Copper Sulphate Anhydrous
- EDTA Dipotassium Salt
- Ferrous Sulfide (1317-37-9)
- Lead Tetroxide (1314-41-6)
- Litharge (1317-36-8) (223.2 g/mol)
- Manganese Dioxide (1313-13-9)
- Phosphogypsum Dihydrate (CaSO4.2H2O)
- Poly(Diallyldimethylammonium Chloride) (26062-79-3) ((C8H16ClN)n)
- Aiocrol (128.50 g/mol) (210 Degree C)
- 2,4-Dinitrophenylhydrazine (119-26-6) (C6H6N4O4)
- Ammonium Pentaborate (B5H4NO8)
- Hydrofluoric Acid 60%
- Hydrofluoric Acid 70%
- Benzyl Bromide (100-39-0)
- Ferric Chloride Liquid
- Sodium Sulphide Hydrate Flakes
- Bentonite Powder
- Caustic Soda Lye
- Methyl Succinyl Chloride
- Aluminium Trihydroxide (21645-51-2)
- Titanium Ethoxide (3087-36-3 )
- Allyl Bromide (106-95-6)
- Copal Gum (9000-14-0)
- Raw Starch
- Phenyl (2396-01-02) (C6H5)
- Cationic Polyelectrolyte Powder
- Wacker L097 (178.50 g/mol)
- Sodium Metatitanate (12034-36-5)
- Precipitated Silica (60.08 g/mol)
- Iron Oxide (1309-37-1)
- Red Phosphorus (7723-14-0)
- Silicon Carbide (409-21-2)
- Chromotropic Acid Disodium Salt
- Copper Pyrithione (154592-20-8)
- 4-Hydrazinobenzoic Acid (619-67-0)
- Roasted Molybdenum Concentrate
- Sodium Aluminate (11138-49-1)
- Selenium Dioxide (7446-08-4)
- Lithopone (1345-05-07) (BaO4S2Zn)
- Gadolinium Oxide (12064-62-9)
- Kaolin (1332-58-7) (Al2H4O9Si2)
- Potassium Borohydride (13762-51-1)
- Quinizarin (81-64-1) (C14H8O4)
- Soda Lime (8006-28-8)
- Polyelectrolyte (150000 g/mol) (350 Degree C)
- Arsenic Trioxide (1327-53-3)
- Safolite (210.0 g/mol) (320 Degree C)
- N-Heptane 99% (100.205 g/mol)
- Kamicryl 400 (350.00 g/mol)
- TCF-10 (312.56 g/mol) (220 Degree C)
- Sunflower Acid oil
- 3,3-Dichlorobenzidine (91-94-1) (C12H10Cl2N2)
- Sodium Bisulfate Monohydrate
- Oxone (614.7 g/mol) (300 Degree C)
- Formaldehyde Sodium Sulfoxylate Dihydrate
- Calcined Alumina (101.96 g/mol)
- PPG-2000 ((C3H6O)nH2O) (2000 g/mol)
- 3-Bromoanisole (2398-37-0) (C7H7BrO)
- LO Acid (180.25 g/mol)
- Lead Naphthenate (61790-14-5)
- White Spirit (140.00 g/mol)
- Tin Chloride Anhydrous
- Heptanoic Anhydride (626-27-7)
- Palladium on Activated Carbon 5%
- 2,2-Dimethoxypropane (77-76-9) (C5H12O2)
- Polyglycerol Polyricinoleate
- EDTA Zinc (C10H12N2Na2O8Zn)
- Sodium Metavanadate (13718-26-8)
- Sodium Molybdate Anhydrous
- Sodium Oxalate (62-76-0)
- Sodium Tungstate (10213-10-2)
- Stannic Oxide (18282-10-5)
- Strontium Chromate (7789-06-02)
- Thiourea Dioxide (1758-73-2)
- Strontium Sulphate (7759-02-06)
- Titanium Oxychloride (92344-13-3)
- Vanadium Pentoxide (1314-62-1)
- Yttrium Oxide (1314-36-9)
- Tungstic Acid (7783-03-01)
- Zinc Chromate (13530-65-9)
- Zinc Formaldehyde Sulfoxylate
- Zinc Lactate (16039-53-5)
- Zinc Silicofluoride (16871-71-9)
- Polyaluminium Chloride (1327-41-9)
- Strontium Aluminate (12004-37-4)
- Potassium Periodate (7790-21-8)
- Potassium Fluorotitanate (K2TiF6)
- Pyrophyllite (Al2Si4O10(OH)2) (360.31 g/mol)
- Silicon Nitride (12033-89-5)
- Sodium Bentonite (85049-30-5)
- Tungsten Carbide (12070-12-1)
- Antimony Trisulphide (1345-04-06)
- Trimethylsulfoxonium Bromide (25596-24-1)
- Calcium Borate (12007-56-6)
- Cuprous Chloride (7758-89-6)
- Sodium Tolyltriazole (64665-57-2)
- Hexafluorozirconic Acid (12021-95-3)
- 2,5-Dimethoxyaniline (102-56-7) (C8H11NO2)
- 1,4-Dithiane-2,5-Diol (40018-26-6) (C4H8O2S2)
- Calcium Bentonite (404.91 g/mol)
- Potassium Tetrafluoroaluminate (14484-69-6)
- Alcohols C9-11-Iso- C10-Rich Ethoxylated
- Linseed Oil (880 g/mol)
- Chrome Oxide Green
- Mixed Xylene (C8H10)
- Bentonite (1302-78-9) (Al2H2O12Si4)
- Magnesium Acetate (142-72-3)
- Sodium Formaldehyde Sulfoxylate
- Calcium Nitrite (13780-06-08)
- Sodium Stannate (12209-98-2)
- Aniline (62-53-3) (C6H5NH2)
- N,N-Dimethyl aniline (121-69-7)
- 1-Ethoxy-2-Propanol (1569-02-04) (C5H12O2)
- Formaldehyde (50-00-0) (CH2O)
- Silicone Defoamer (800 g/mol)
- Aluminium Sulphate Anhydrous
- 2,2,2-Trichloroethanol (115-20-8) (C2H3Cl3O)
- Benzeneseleninic Anhydride (17697-12-0)
- Calcite Powder (100.086 g/mol)
- Valeronitrile (110-59-8) (C5H9N)
- Triethoxymethylsilane (2031-67-6) (C7H18O3Si)
- Ammonium Cerium Nitrate
- Isobutylene oxide (558-30-5)
- Titanium Carbide (12070-08-05)
- Tellurium Dioxide (7446-07-3)
- Styrene (100-42-5) (C8H8)
- 3-Chloro-2-Methylaniline (87-60-5) (C7H8ClN)
- Potassium Peroxymonosulfate Triple Salt
- Sodium Acid Pyrophosphate
- 48-R (212.30 g/mol) (320 C)
- Fumed Silica (60.08 g/mol)
- Spodumene Concentrate (186.09 g/mol)
- 2-Ethoxyethanol (110-80-5) (C4H10O2)
- Lawesson's Reagent (19172-47-5)
- Methyl Silicate (681-84-5)
- Dialdehyde Starch (9047-50-1)
- Bromine (7726-95-6) (Br2)
- 2-Ethylhexanoic Acid (149-57-5)
- Barium sulfide (21109-95-5)
- Benzene (71-43-2) (C6H6)
- Nitromethane (75-52-5) (CH3NO2)
- Chloroform (67-66-3) (CHCl3)
- Decolite DP (CH3NaO3S)
- Stearylamine (124-30-1) (C18H39N)
- Strontium Carbonate (1633-05-2)
- Triethylamine (121-44-8) (C6H15N)
- De foamer
- Methyl Linoleate (112-63-0)
- 1-Naphthol (90-15-3) (C10H8O)
- 1,2-Epoxybutane (106-88-7) (C4H8O)
- Butylated Hydroxytoluene (128-37-0)
- Carbitol (111-90-0) (C6H14O3)
- Iodine Monochloride (7790-99-0)
- Hexamine (100-97-0)
- 1-Hydroxyethylidene-1,1-Diphosphonic Acid (2809-21-4)
- Lignin (94113-57-2) (C18H13N3Na2O8S2)
- Nitroethane (79-24-3) (C2H5NO2)
- Ferrous Chloride (7758-94-3)
- Para Nitrochlorobenzene (100-00-5)
- Perchloroethylene (127-18-4) (C2Cl4)
- Phenidone (92-43-3) (C9H10N2O)
- Methyl Cyanoacetate (105-34-0)
- Potassium Bifluoride (7789-29-9)
- Methylal (109-87-5) (C3H8O2)
- Aluminum Sulfate Non Ferric
- Calcium Sulfate (7778-18-9)
- Dimethyl Sulphate (77-78-1)
- EDTA Disodium Salt
- Ethyl Silicate (78-10-4)
- Fluosilicic Acid (16961-83-4)
- Polyvinyl Alcohol (9002-89-5)
- Potassium Dichromate (7778-50-9)
- Potassium Silico Fluoride
- Sodium Borohydride (16940-66-2)
- Sodium Hydrosulfite (7775-14-6)
- Sodium Lignosulfonate (8061-51-6)
- Sodium sulfite (7757-83-7)
- Stannous Chloride (Powder)
- Sulfamic Acid (5329-14-6)
- Toluene (108-88-3) (C6H5CH3)
- Tert-Butyl Nitrite (540-80-7)
- Triethyl Orthoformate (122-51-0)
- Zeolites (1318-02-01) (Aluminosilicate)
- 1,1,2,2-Tetrachloroethane (79-34-5) (C2H2Cl4)
- Sodium Hypophosphite (10039-56-2)
- Sodium Hypophosphite
- Magnesium Silicofluoride (16949-65-8)
- Calcium aluminate (12042-68-1)
- Calcium Lignosulphonate (8061-52-7)
- 4-Chlorobenzotrifluoride (98-56-6) (C7H4ClF3)
- Acetone (67-64-1) (C3H6O)
- Barium Sulphate (7727-43-7)
- Aluminium Oxide (1344-28-1)
- Ammonium Oxalate (1113-38-8)
- Benzotriazole (95-14-7) (C6H5N3)
- N,N-Dimethylethanolamine (108-01-0) (C4H11NO)
- Nitrocellulose (9004-70-0) ((C6H7N3O11)n)
- Phosphorus Tribromide (7789-60-8)
- Potassium Hydrogen Phthalate
- Potassium Hydroxide (1310-58-3)
- Methyltrimethoxysilane (1185-55-3) (C4H12O3Si)
- Potassium Titanium Fluoride
- N-Propyl Acetate (109-60-4)
- Propylene Glycol Monomethyl Ether Acetate
- 4-Methyl-2-Pentanol (108-11-2) (C6H14O)
- Aluminium Chloride (7446-70-0)
- Aluminium Trifluoride (AlF3)
- Copper Oxide (1317-38-0)
- Diethyl Ether (60-29-7)
- Diethylene Glycol (111-46-6)
- Furfuryl Alcohol (98-00-0)
- Ethylenediamine (107-15-3) (C2H8N2)
- Glyoxal (107-22-2) (C2H2O2)
- Gum Rosin (8050-09-7)
- Ferric Sulfate (10028-22-5)
- Hydrobromic Acid (10035-10-6)
- Formamide (75-12-7) (CH3NO)
- Methylcyclohexane (108-87-2) (C7H14)
- Potassium Ferricyanide (13746-66-2)
- Monoethanolamine (141-43-5) (C2H7NO)
- Potassium Fluoborate (14075-53-7)
- N-Methyl Pyrrolidone (872-50-4)
- Propylene Carbonate (108-32-7)
- Resist Salt (C6H4NNaO5S)
- Sodium Carbonate (497-19-8)
- Sodium Persulfate (7775-27-1)
- Sodium Thiocyanate (540-72-7)
- Tetrabutylammonium Bromide (1643-19-2)
- Tetraisopropyl Titanate (546-68-9)
- Sulfuric Acid (7664-93-9)
- Triacetin (102-76-1) (C9H14O6)
- Trichloroethylene (79-01-6) (C2HCl3)
- Tert-Butyl Chloride (507-20-0)
- Trimethylamine (75-50-3) (C3H9N)
- Lithium Hydroxide Monohydrate
- Methoxy Propanol (107-98-2)
- N-Ethylethanamine (109-89-7) (C4H11N)
- Naphthalene (91-20-3) (C10H8)
- Tetrahydrofuran (109-99-9) (C4H8O)
- Sodium Hexametaphosphate (68915-31-1)
- Sodium Nitrite (7632-00-0)
- Sodium Metaborate (7775-19-1)
- Aluminum fluoride (7784-18-1)
- N-Butyl Bromide (109-65-9)
- Sodium Petroleum Sulfonate
- Sodium Aluminum Silicate
- Dibasic Ester (Dimethyl esters)
- Chlorinated Ethylene Vinyl Acetate
- Fumed Silica Hydrophilic
- Potassium Hexafluorotitanate (16919-27-0)
- Triethanolamine (102-71-6) (149.19 g/mol)
- China Clay (258.16 g/mol)
- Anthracene-9,10-Dicarboxaldehyde (7044-91-9) (C16H10O2)
- N-Butyldiethanolamine (102-79-4) (C8H19NO2)
- N-Ethyldiethanolamine (139-87-7) (C6H15NO2)
- N-Formylmorpholine (4394-85-8) (C5H9NO2)
- Lanthanum Oxide (1312-81-8)
- Ethylene Glycol Monoethyl Ether1
- Manganese Acetate Anhydrous
- 2,2-Diphenyl-1-Picrylhydrazil (13775-53-6) (C18H12N5O6)
- 1-Bromo-3-Chloropropane (109-70-6) (C3H6BrCl)
- 1-Bromo-3-Chloropropane (109-70-6) (C3H6BrCl)
- Pyrogallol (87-66-1) (C6H6O3)
- 1,3-Dichloropropane (142-28-9) (C3H6Cl2)
- Pyrrole (109-97-7) (C4H5N)
- 1,4-Benzoquinone (106-51-4) (C6H4O2)
- Sodium Periodate (7790-28-5)
- Sodium Tert-Butoxide (865-48-5)
- Tert-Butyl Acetate (540-88-5)
- Tetrahydrofurfuryl Alcohol (97-99-4)
- Trifluoroacetic Acid (76-05-1)
- Trimethyl Orthoacetate (1445-45-0)
- Trimethylsulfoxonium Iodide (1774-47-6)
- Zinc Bromide (7699-45-8)
- Sodium Iodate (7681-55-2)
- Diethylene Glycol Monobutyl Ether Acetate
- Diethyl Phosphite (0762-04-09)
- Bromobenzene (108-86-1) (C6H5Br)
- 3-Bromopyridine (626-55-1) (C5H4BrN)
- Phosphomolybdic Acid (12026-57-2)
- Thioacetamide (62-55-5) (C2H5NS)
- Butyl lactate (138-22-7)
- 2-Bromopropane (75-26-3) (C3H7Br)
- 2-Chlorophenol (95-57-8) (C6H5ClO)
- Methallyl Chloride (563-47-3)
- 2-Chloropyridine (0109-09-01) (C5H4ClN)
- N,N-Dimethylcyclohexylamine (98-94-2) (C8H17N)
- Lithium Sulphate (10377-48-7)
- 2-Pyrrolidone (616-45-5) (C4H7NO)
- Copper Glycinate (13479-54-4)
- 2,4-Dichlorobenzoic Acid (50-84-0)
- Diethylene Glycol Dimethyl Ether
- 2-Hydroxypropyl Acrylate (999-61-1)
- 2,6-Dichlorophenol (87-65-0) (C6H4Cl2O)
- Ammonium Iodide (12027-06-04)
- 1,2-Dimethoxyethane (110-71-4) (C4H1002)
- Pyruvic Acid (127-17-3)
- Barium Hydroxide Octahydrate
- Benzenesulphonamide (98-10-2) (6H7NO2S)
- Trifluoromethanesulfonic Acid (1493-13-6)
- Benzoin (119-53-9) (C14H12O2)
- Bromoacetyl Bromide (598-21-0)
- Chlorobenzene (108-90-7) (C6H5Cl)
- Chromium Trioxide (1333-82-0)
- Copper Iodide (7681-65-4)
- Diisopropyl Ether (108-20-3)
- Zinc Iodide (10139-47-6)
- N-Iodosuccinimide (0516-12-1) (C4H4INO2)
- Diallyldimethylammonium Chloride (7398-69-8)
- 6-Chloro-2-Hexanone (10226-30-9) (C6H11ClO)
- Fluorobenzene (0462-06-06) (C6H5F)
- N,O-Bis Trimethylsilyl Acetamide (10416-59-8)
- Isobutyryl Chloride (79-30-1)
- N,N-Dimethylethylenediamine (110-70-3) (C4H12N2)
- Sodium Bis(Trimethylsilyl)Amide (1070-89-9)
- Turpentine (9005-90-7) (136.24 g/mol)
- Dimethyl Carbonate (616-38-6)
- Dimethyl Malonate (108-59-8)
- Dimethylacetamide (127-19-5) (C4H9NO)
- Ethyl Chloroformate (541-41-3)
- Ethyl Cyanoacetate (105-56-6)
- Hexylresorcinol (136-77-6) (C12H18O2)
- Iodoform (75-47-8) (CHI3)
- Isatoic Anhydride (118-48-9)
- 2,4-Dinitrochlorobenzene (97-00-7) (C6H3ClN2O4)
- Lithium Amide (7782-89-0)
- Malononitrile (109-77-3) (C3H2N2)
- Methyl Amyl Ketone
- Methyl Hydroxyethyl Cellulose
- 1,3-Dibromopropane (109-64-8) (C3H6Br2)
- N,N,4-Trimethylaniline (99-97-8) (C9H13N)
- P-Toluidine (106-49-0) (C7H9N)
- Manganese Chloride Tetrahydrate
- Pelargonic Acid (112-05-0)
- Potassium Formate (590-29-4)
- 2-Nitrochlorobenzene (88-73-3) (C6H4ClNO2)
- Potassium Tert-Butoxide (865-47-4)
- Propargyl Alcohol (107-19-7)
- Alpha-Toluenesulfonyl Chloride (1939-99-7)
- Sodium Silicate Liquid
- Nickel Acetate Tetrahydrate
- Ammonium Polyphosphate Liquid
- Phosphoric Acid 85%
- 1-Bromopentane (110-53-2) (C5H11Br)
- Sodium Hypophosphite Monohydrate
- Nitrocellulose 30/50 (Powder)
- Nitrocellulose 10/25
- Sodium Hydroxide Prills
- Propyltrimethoxysilane (1067-25-0) (C6H16O3Si)
- Acetone LR (C3H6O)
- Barium Hydroxide Monohydrate
- Isopropyl Alcohol LR
- Ferric Acetylacetonate (14024-18-1)
- 2-Methoxy-4-Nitroaniline (97-52-9) (C7H8N2O3)
- Triethyl Orthoacetate (78-39-7)
- 5-Nitroisophthalic Acid (618-88-2)
- Crude Soybean Oil
- Sodium Ferrocyanide Decahydrate
- Mix Xylene (106.16 g/mol)
- Aminoethylpiperazine (140-31-8) (C6H15N3)
- Potassium Hydroxide Flakes
- Potassium Carbonate Granules
- 3-Aminophenol (591-27-5) (C6H7NO)
- 4-Chloroaniline (106-47-8) (C6H6ClN)
- Silver Iodide (7783-96-2)
- 4-Chlorobenzoic Acid (74-11-3)
- Ferric Chloride Hexahydrate
- Glyoxal 40% (C2H2O2)
- Methyl Lactate (547-64-8)
- 2-Bromo-4-Methoxyacetophenone (2632-13-5) (C9H9BrO2)
- Sodium Thiosulfate Pentahydrate
- 1-Bromo-4-Chlorobutane (6940-78-9) (C4H8BrCl)
- Sulfanilic Acid (121-57-3)
- Tert-Butyldimethylsilyl Chloride (18162-48-6)
- 1-Phenylethylamine (618-36-0) (C8H11N)
- Tetra-N-Butyl Titanate (5593-70-4)
- 1,1,3,3-Tetramethyldisiloxane (3277-26-7) (C4H12OSi2)
- Tetradecane (629-59-4) (C14H30)
- 1,3-Dichloroacetone (0534-07-06) (C3H4Cl2O)
- Tributyltin Chloride (1461-22-9)
- 1,3-Dichlorobenzene (541-73-1) (C6H4Cl2)
- Trichloroethane (71-55-6) (C2H3Cl3)
- Triethoxysilane (998-30-1) (C6H16O3Si)
- 2-Chloro-N,N-Diethylethylamine Hydrochloride (869-24-9)
- Triethyl Phosphate (78-40-0)
- Trimethyl Phosphate (512-56-1)
- Gold Chloride (13453-07-01)
- 2-Chlorobenzoyl Chloride (609-65-4)
- 2-Hydroxypyridine (0142-08-05) (C5H5NO)
- 2-Nitrobenzaldehyde (552-89-6) (C7H5NO3)
- 2,2,2-Trifluoroethanol (75-89-8) (C2H3F3O)
- Boron Trifluoride (7637-07-2)
- 2,2,6,6-Tetramethylpiperidine 1-Oxyl (2564-83-2) (C9H18NO)
- Bromochloromethane (74-97-5) (CH2BrCl)
- 3-Methoxypropylamine (5332-73-0) (C4H11NO)
- 2,3-Dihydrofuran (1191-99-7) (C4H6O)
- 4-Nitrobenzoic Acid (62-23-7)
- 3-Chloropropionyl Chloride (625-36-5)
- Tripropylamine (102-69-2) (C9H21N)
- 3-Methoxy-1-Propanol (1589-49-7) (C4H10O2)
- Phosphorus Pentachloride (10026-13-8)
- Potassium Ethyl Xanthate
- Ethyl Isonipecotate (1126-09-06)
- 3-Methylamino Propylamine (6291-84-5)
- M-Anisidine (536-90-3) (C7H9NO)
- Indoline (496-15-1) (C8H9N)
- Lithium Sulfate (10102-25-7)
- Ammonium Bisulphate (7803-63-6)
- Antimony Trichloride (10025-91-9)
- Hexafluoroisopropanol (920-66-1) (C3H2F6O)
- Isoamyl Bromide (107-82-4)
- Bauxite (1318-16-7) (Al2H2O4)
- 4-Methoxyphenylacetonitrile (104-47-2) (C9H9NO)
- Benzonitrile (100-47-0) (C7H5N)
- Benzyltrimethylammonium Chloride (56-93-9)
- Methyl Abietate (127-25-3)
- Bromocyclopentane (137-43-9) (C5H9Br)
- Cadmium Selenide (1306-24-7)
- Methyl 3-aminocrotonate (14205-39-1)
- Calcium Hexaboride (12007-99-7)
- Carbon Tetrabromide (558-13-4)
- Cesium Carbonate (534-17-8)
- 2-Propylheptanol (10042-59-8) (C10H22O)
- 2-Phosphonobutane-1,2,4-Tricarboxylic Acid (37971-36-1)
- 4-2-Hydroxyethyl Morpholine (622-40-2)
- Caffeic Acid (331-39-5)
- Boron Trichloride (10294-34-5)
- Sodium Aluminium Phosphate
- Sodium Molybdate Dihydrate
- Oxalyl Chloride (79-37-8)
- Vinyl Tris-Methyl Ethyl Ketoxime Silane
- Potassium Pyrosulfate (7790-62-7)
- Sodium Glutamate Monohydrate
- 2-Chloroethylamine Hydrochloride (870-24-6)
- 1,6-Dibromohexane (0629-03-08) (C6H12Br2)
- 1,5-Pentanediol (111-29-5) (C5H12O2)
- Cyclohexene Oxide (286-20-4)
- Diethyl-M-Aminophenol (91-68-9) (C10H15NO)
- N,N,N',N'-Tetramethylethylenediamine (110-18-9) (C6H16N2)
- Trimethylsilylacetylene (1066-54-2) (C5H10Si)
- Perchloric Acid (7601-90-3)
- 2-Methylnaphthalene (91-57-6) (C11H10)
- Cesium Nitrate (7789-18-6)
- Methoxyamine Hydrochloride (593-56-6)
- Isooctane (540-84-1) (C8H18)
- Aluminum Dihydrogen Phosphate
- Bis(2-Ethylhexyl)Amine (106-20-7) (C16H35N)
- Isophthaloyl Chloride (99-63-8)
- 2-Bromofluorobenzene (1072-85-1) (C6H4BrF)
- Ortho-Phthalaldehyde (643-79-8) (C8H6O2)
- P-Hydroxybenzyl Cyanide (14191-95-8)
- Methyl Diethanolamine (105-59-9)
- 3-Aminobenzonitrile (2237-30-1) (C7H6N2)
- Magnesium Oxide 85%
- Pentasodium Diethylenetriaminepentaacetate (0140-01-02)
- 3-Nitrobenzaldehyde (99-61-6) (C7H5NO3)
- Hyflo Supercel (Hyflo)
- Ammonium Alum Lumps
- Chloroacetonitrile (107-14-2) (C2H2ClN)
- Cyclopentyl Methyl Ether
- Diethyl Chlorophosphate (814-49-3)
- Aluminium Formate (7360-53-4)
- Orthosilicic Acid (10193-36-9)
- Diethyl Oxalate (95-92-1)
- Sodium Sulfide Flakes
- Glucuronolactone (32449-92-6) (C6H8O6)
- Hematoxylin (517-28-2) (C16H14O6)
- Isonicotinic Acid (55-22-1)
- Lithium Diisopropylamide (4111-54-0)
- Methyltrioctylammonium Chloride (5137-55-3)
- Molybdic Acid (7782-91-4)
- Monoisopropanolamine (78-96-6) (C3H9NO)
- N-Decane (124-18-5) (C10H22)
- P-Anisidine (104-94-9) (C7H9NO)
- Phenyl Isothiocyanate (103-72-0)
- R-1-phenylethylamine (3886-69-9) (C8H11N)
- Sodium Cyanoborohydride (25895-60-7)
- Sodium Monochloro Acetate
- Sodium Permanganate Anhydrous
- 4-Bromobenzoic Acid (586-76-5)
- 2,4,6-Tribromophenol (118-79-6) (C6H3Br3O)
- 4-Chloro-3-Nitrobenzoic Acid (96-99-1)
- 4-Carboxybenzaldehyde (619-66-9) (C8H6O3)
- 4-Chloro-O-Phenylenediamine (95-83-0) (C6H7ClN2)
- Alpha-Amylase (9000-90-2) (C15H17N3OS2)
- 2,2-Dimethoxy-2-Phenylacetophenone (24650-42-8) (C16H16O3)
- N-Ethylpiperazine (5308-25-8) (C6H14N2)
- 8-Amino-1,3,6-Naphthalenetrisulfonic Acid (117-42-0)
- 1-Bromonaphthalene (90-11-9) (C10H7Br)
- Acetaldoxime (107-29-9) (C10H10N2O)
- 2,6-Dihydroxybenzoic Acid (0303-07-01)
- Anionic Polyelectrolyte (C6H4N8O4)
- Potassium Peroxymonosulfate (10058-23-8)
- 2,3-Epoxypropyltrimethylammonium Chloride (3033-77-0)
- Non Ferric Alum Slab
- 2,4,5-Trichloroaniline (636-30-6) (C6H4Cl3N)
- Non Ferric Alum Powder
- Polyvinyl Alcohol 2488
- Cyanoacetic Acid 70% Solution
- 3-Chloroperoxybenzoic Acid (937-14-4)
- Sodium Hydroxide Flakes Industrial Grade
- 2-Methylaniline (95-53-4) (C7H8FN)
- Hydrogen Peroxide Industrial Grade
- N-Pentyl Amine (110-58-7)
- Jeffamine D-230 (230 g/mol)
- Calcined Alumina Powder
- N-Octyl Bromide (111-83-1)
- N-Hexyl Bromide (111-25-1)
- Zircon Flour (ZrO4Si)
- 5,5-Dimethylhydantoin (77-71-4) (C5H8N2O2)
- Germanium Dioxide (1310-53-8)
- 5-Iodo-2-Methylbenzoic Acid (54811-38-0)
- Ethyl Iodide (75-03-6)
- 3-Ethoxypropylamine (6291-85-6) (C5H13NO)
- 5-Bromo-2,4-Dichloropyrimidine (36082-50-5) (C4HBrCl2N2)
- 2,5-Di-Tert-Butylhydroquinone (88-58-4) (C14H22O2)
- 4-(2,3-Epoxypropoxy)Carbazole (51997-51-4) (C15H13NO2)
- Lithium Acetate Anhydrous
- Lithium Aluminium Hydride
- Arsenic Triiodide (7784-45-4)
- 2-Pentyn-1-Ol (6261-22-9) (C5H8O)
- Titanium Dioxide Rutile 6618
- Isopar G (Liquid)
- Sodium Formaldehyde Bisulfite
- Sodium Hydrosulfide 30% Liquid
- Demulsifier (680.40 g/mol) (210 Degree C)
- Acetaldehyde 30% (CH3CHO)
- Quartz Silica (14808-60-7)
- Formic Acid 85 %
- Octadecyltrichlorosilane (0112-04-09) (C18H37Cl3Si)
- 2,4-Dichlorobenzyl Chloride (94-99-5)
- Ethyl D-Lactate (7699-00-5)
- Trimethylammonium Chloride (593-81-7)
- Gum Copal Dust
- Sodium Polysulfide (1344-08-07)
- Ammonium Paratungstate (11120-25-5)
- Delta-Valerolactone (542-28-9) (C5H8O2)
- Lime Powder (Ca(OH)2)
- Bismuth Nitrate Pentahydrate
- Triphenylphosphine Oxide (791-28-6)
- Copper 12% EDTA
- Diethyl Sulfate (64-67-5)
- 4-Methoxy-2-Methyldiphenylamine (41317-15-1) (C14H15NO)
- SLACK WAX (420 g/mol)
- Calcined Powder (60.08 g/mol)
- Refined Palm Oil
- 1-Bromohexadecane (112-82-3) (C16H33Br)
- Isobutyl Triethylamine (Liquid)
- 4-Chlorobenzenesulfonyl Chloride (98-60-2)
- Phenyl Chloroformate (1885-14-9)
- N-Propyl Bromide (106-94-5)
- Tetrabutylammonium Hydrogen Sulfate
- Nitric Acid 60%
- Trisodium Citrate Dihydrate AR
- Polyvinyl Alcohol Powder
- Acrylic Polymer (Solid)
- Sodium Ethoxide Powder
- Zinc Fluoride Anhydrous
- Glyoxylic Acid 50%
- Nitrocellulose Cotton (Powder)
- Polyetheramine (Liquid) (1500 g/mol)
- Refined Naphthalene (Solid)
- Phosphoric Acid 85% Industrial Grade
- Phosphotungstic Acid Anhydrous
- Graphene Oxide Powder
- Lead Chromate (7758-97-6)
- 1,5-Diaminonaphthalene (2243-62-1) (C10H10N2)
- Ferric Ammonium Oxalate
- Lead Fluoborate (13814-96-5)
- Zinc Selenite (13597-46-1)
- Nitrocellulose HX 8/13
- Trimethylsulfonium Iodide (2181-42-2)
- Disodium Hydrogen Phosphate Anhydrous AR
- Sulfuric Acid LR
- Oleylamine, Ethoxylated (26635-93-8)
- Potassium Hexafluorophosphate (17084-13-8)
- Sodium Aluminium Sulfate Anhydrous
- Calcined Petroleum Coke
- 1-(3-Dimethylaminopropyl)-3-Ethylcarbodiimide Hydrochloride (25952-53-8)
- Mucic Acid (526-99-8)
- Naphthenic Oil (350 g/mol)
- Phosphoric Acid LR
- 3-Nitrophthalic Acid (0603-11-2)
- Di(Propylene Glycol) Methyl Ether Acetate
- 4-Nitrophthalic Acid (610-27-5)
- 1-Bromo-4-Iodobenzene (589-87-7) (C6H4BrI)
- 3,5-Dinitrosalicylic Acid (609-99-4)
- N-Propyl Iodide (0107-08-04)
- Triethyl Borate (150-46-9)
- Aerosil 200 (Powder)
- Sodium Pentaborate (12007-92-0)
- Zeolite Powder (2191 g/mol)
- Tin Chloride Dihydrate
- Quicklime Crystal (CaO)
- 1-Phenoxy-2-Propanol (770-35-4) (C9H12O2)
- 4-Methoxybenzylamine (2393-23-9) (C8H11NO)
- Thinner (142.5 g/mol) (150 Degree C)
- Hexamethylphosphoramide (680-31-9) (C6H18N3OP)
- 2-Butyn-1-Ol (0764-01-02) (C4H6O)
- Beta-Cyclocitral (432-25-7) (C10H16O)
- 5-Bromoindole (10075-50-0) (C8H6BrN)
- Crotonic Acid (107-93-7)
- Magnesium Methoxide (109-88-6)
- Tungsten Trioxide (1314-35-8)
- Sodium Chlorite 30% Solution
- Paraformaldehyde 96% (Powder)
- Glyceryl Trioleate (122-32-7)
- Liquid Soya Lecithin
- 1,3-Dibromo-5,5-Dimethylhydantoin (77-48-5) (C5H6Br2N2O2)
- Lithium Stearate (4485-12-5)
- Tor 3130 (238.25 g/mol)
- Ball Clay (258.16 g/mol)
- 2-2-Methoxyethoxy Ethanol (111-77-3)
- Mono Methyl Ethanolamine
- 4-Nonylphenol Diethoxylate (9016-45-9)
- Distilled Soya Fatty Acid
- M` Swassolve Acetate (110-49-6)
- Swassolve Acetate (111-15-9)
- Diethylene Glycol Monobutyl Ether
- Methoxy Propyl Acetate
- Monoethylamine (75-04-7) (C2H5NH2)
- N-Butylamine (109-73-9) (C4H9NH2)
- Tris-2-Ethylhexyl Amine (1860-26-0)
- 2-Aminoethoxyethanol (0929-06-06) (C4H11NO2)
- Phenyltrimethylammonium Chloride (138-24-9)
- Lithium Bromide Powder
- Tetrapropylammonium bromide (1941-30-6)
- Lithium Chloride Powder
- Lithium Molybdate Powder
- 1,10-Dibromodecane (4101-68-2) (C10H20Br2)
- 1,12-Dibromododecane (3344-70-5) (C12H24Br2)
- 1,4-Dibromo-2,5-Difluorobenzene (327-51-5) (C6H2Br2F2)
- 1,8-Dibromooctane (4549-32-0) (C8H16Br2)
- 10-Bromoundecanoic Acid (18294-93-4)
- 1-Amino-2,4-Dibromoanthraquinone (81-49-2) (C14H7Br2NO2)
- 1-Bromo-2,4-Dichlorobenzene (1193-72-2) (C6H3BrCl2)
- 2-Bromodecanoic Acid (2623-95-2)
- 2-Bromohexanoic Acid (0616-05-07)
- 3-Bromopropyltrimethylammonium Bromide (3779-42-8)
- 2-Bromopropanoic Acid Sodium Salt
- 4-Fluorophenol (371-41-5) (P-Fluorophenol)
- 6-Bromohexyltrimethylammonium Bromide (32765-81-4)
- 7-Bromomethylpentadecane (52997-43-0) (C16H33Br)
- Lithium Bis Fluorosulfony Imide
- Lithium Tetrahydridoborate (16949-15-8)
- Lithium Tri-Tert-Butoxyaluminum Hydride (114772-54-2)
- Benzyl Tri Butyl Ammonium Chloride
- Cis-Bromo Benzoate (61397-56-6)
- 1,1,2-Tribromoethane (78-74-0) (266.76 g/mol)
- 1,2,3-Tribromopropane (96-11-7) (C3H5Br3)
- 1,2-Dibromocyclohexane (5401-62-7) (241.95 g/mol)
- 1,2-Dibromocyclopentane (10230-26-9) (C5H8Br2)
- 1,3-Cyclohexadiene (592-57-4) (C6H8)
- 1,3-Dibromo-2,2-Dimethylpropane (5434-27-5) (C5H10Br2)
- 1,3-Dichloro-2,2-Dimethylpropane (29559-55-5) (C5H10Cl2)
- 1,4-Dibromobenzene (106-37-6) (235.90 g/mol)
- 1,5-Dichloropentane (628-76-2) (C5H10Cl2)
- 10-Bromodecanol (53463-68-6) (C10H21BrO)
- 1-Bromo-6-Chlorohexane (6294-17-3) (C6H12BrCl)
- 1-Bromo-6-Methylheptane (52648-04-01) (199.53 g/mol)
- 1-Bromo-8-Chlorooctane (28598-82-5) (C8H16BrCl)
- 1-Bromoheptane (0629-04-09) (N-Heptyl Bromide)
- 1-Chloro-2-Ethylhexane (0123-04-06) (C8H17Cl)
- 1-Iododecane (2050-77-3) (C10H21I)
- 2,2-Dibromopropane (594-16-1) (201.89 g/mol)
- 2-Bromo-2-Methylpropane (507-19-7) (Tert-Butyl Bromide)
- 2-Bromo-2-Methylpropionylbromide (20769-85-1) (2-Bromo Iso-Butyryl Bromide)
- Phenyllithium (591-51-5) (C6H5Li)
- 4-Tert-Butylphenylmagnesium Bromide (63488-10-8)
- 4-Bromobenzyl Bromide (589-15-1)
- 4-Bromobutyl Acetate (4753-59-7)
- 6-Bromohexyl Acetate (68797-94-4)
- 6-Bromo-1-Hexene (2695-47-8) (163.06 g/mol)
- 7-Bromo-1-Heptene (03-09-4117) (C7H13Br)
- 8-Bromooctanol (50816-19-8) (C8H17BrO)
- Allyltrimethylsilane (762-72-1) (C6H14Si)
- Bromo Acetonitrile (590-17-0)
- Bromocyclohexane (108-85-0) (Cyclohexyl Bromide)
- Calcium Bromide Solution 52%
- Cyclopropyl Methyl Bromide
- Ethyl 2-Bromohexanoate (615-96-3)
- Ethyl 7-Bromoheptanoate (29823-18-5)
- Ethyl-2-Bromopentanoate (615-83-8) (C7H13BrO2)
- Ethyl 6-Bromo-2,2-Dimethylhexanoate (78712-62-6) (C10H19BrO2)
- Ethyl-8-Bromooctanoatee (29823-21-0) (1-Acetoxy-6-Bromohexane)
- Iso-Propyl Magnesium Chloride Lithium Chloride Complex
- Methyl 6-Bromohexanoate (14273-90-6)
- Methyl-2-Bromopentanoate (19129-92-1) (C6H11BrO2)
- N-Hexyl Magnesium Bromide 2m In Thf
- Tetrabutylammonium Hydroxide 40% Solution
- Benzalkonium Chloride 80 % Solution
- Hydrobromic Acid 33% In Glacial Acetic Acid
- Hydrobromic Acid 62% Solution
- Sodium Bromide 45% Solution
- Calcium Bromide 52% Solution
- Lithium Bromide 55% Solution
- Phenylmagnesium Bromide 2m In Thf
- Phenylmagnesium Chloride 2m In Thf
- Ethyltriphenylphosphonium Bromide (1530-32-1)
- Tetrabutylammonium Iodide (311-28-4)
- Diisopropylethylamine (7087-68-5)
- PEG-150 Distearate (Solid)
- Sodium Methoxide Powder
- Ethyl Bromide (74-96-4)
- 1,4-Dibromobutane (110-52-1) (Tetramethylene Dibromide)
- Sodium Bromide Powder
- 3-Bromonitrobenzene (585-79-5) (1-Bromo-3-Nitrobenzene)
- Lithium 12-Hydroxystearate (7620-77-1)
- 1,10-Dichlorodecane (2162-98-3) (Liquid)
- 1,4-Dichlorobutane (110-56-5) (Liquid)
- 1,5-Dibromopentane (111-24-0) (Pentamethylene Dibromide)
- 1-Bromo-2-Chloroethane (107-04-0) (Liquid)
- 1-Bromooctadecane (112-89-0) (Solid)
- Sodium Bromide Solution 40%
- Tert-Butylmagnesium Chloride 2m In Thf
- 3-Chloro-1-Propanol (627-30-5) (Liquid)
- 4-Bromo-1-Butene (5162-44-7) (Liquid)
- 1-Bromo-4-Fluorobenzene (460-00-4) (Liquid)
- 5-Bromo-1-Pentene (1119-51-2) (Liquid)
- 2-Ethylhexyl Bromide (18908-66-2)
- 1-Bromo-5-Chloropentane (54512-75-3) (Liquid)
- Ethyl Bromide (74-96-4)
- Ethyl Magnesium Chloride 2M In THF
- Personal & Home Care
- Citric Acid Anhydrous
- Cetyltrimethylammonium Bromide (57-09-0)
- Glyoxylic Acid (0298-12-4)
- Sodium Sulphate (7757-82-6)
- 2-phenoxyethanol (122-99-6) (C8H10O2)
- Benzenesulphonic Acid (98-11-3)
- Picramic Acid (96-91-3)
- Ethylene Glycol Distearate
- 1-Tridecanol (112-70-9) (C13H28O)
- Sodium Carbonate Decahydrate
- Alcohol Ethoxylate
- Aluminium Chlorohydrate (12359-72-7)
- Azelaic Acid (123-99-9)
- Benzoyl Peroxide (94-36-0)
- Calcium Thioglycolate (814-71-1)
- Capric Acid (334-48-5)
- Castor Oil (8001-79-4)
- Citric Acid Anhydrous
- Citric Acid Monohydrate
- Cocoamine (Amines) (Liquid)
- Dehydroacetic Acid (520-45-6)
- Denatonium Saccharide (90823-38-4)
- Dimethylpolysiloxane (9016-00-6) ((C2H6OSi)n)
- Emulsifying Wax (Nonionic self-emulsifying wax)
- Hexylene Glycol (107-41-5)
- Hydrogen Peroxide (7722-84-1)
- Hydrogenated Castor oil
- Hydroquinone (123-31-9) (C6H6O2)
- Isopropyl Palmitate (142-91-6)
- Lactic Acid (50-21-5)
- Lanolin (8006-54-0) (Adeps lanae)
- Lauryl Glucoside (59122-55-3)
- Lauryl Trimethyl Ammonium Chloride
- Methylparaben (99-76-3) (C8H8O3)
- Nonoxynol-9 (14409-72-4) (C33H60O10)
- Oleic Acid (112-80-1)
- Oleyl Alcohol (143-28-2)
- Olive Oil (8001-25-0)
- Palm Oil (8002-75-3)
- Palm Stearin (Solid)
- Paraffin Wax (8002-74-2)
- Potassium Stearate (593-29-3)
- Potassium Tripolyphosphate (Powder)
- Propyl Gallate (121-79-9)
- Propylparaben (94-13-3) (C10H12O3)
- Ammonium Hydroxide (1336-21-6)
- Pyrithione Zinc
- Pyrithione Zinc (13463-41-7)
- Resorcinol (108-46-3) (C6H6O2)
- Rice Bran Wax
- Salicylic Acid (69-72-7)
- Silicone Oil (63148-62-9)
- Sodium Percarbonate (15630-89-4)
- Tetrapotassium Pyrophosphate (7320-34-5)
- Tetrasodium Pyrophosphate Anhydrous
- 2-Ethylhexyl Laurate (20292-08-04)
- 2-Ethylhexyl Salicylate (118-60-5)
- 3-O-Ethyl-l-Ascorbic Acid (86404-04-08) (C8H12O6)
- Alcohol Ethoxylates (Fatty Alcohol Ethoxylate)
- Allantoin (97-59-6) (C4H6N4O3)
- Ammonium Lactate (515-98-0)
- Ammonium Thioglycolate (5421-46-5)
- Beeswax (8012-89-3) (Beeswax Absolute)
- Behenyl Alcohol (661-19-8)
- Benzalkonium Chloride (8001-54-5)
- Benzethonium Chloride (121-54-0)
- Benzyl Nicotinate (94-44-0)
- Beta Glucan (9012-72-0)
- Bismuth Oxychloride (7787-59-9)
- Bronopol (52-51-7) (C3H6BrNO4)
- Butylated Hydroxyanisole (25013-16-5)
- Calamine (8011-96-9) (Fe2O4Zn)
- Calcium Hypochlorite (7778-54-3)
- Candelilla Wax (8006-44-8)
- Caprylyl Glycol (1117-86-8)
- Cetrimide (8044-71-1) (C17H38BrN)
- Cetyl Alcohol (36653-82-4)
- Lauroyl Chloride (112-16-3)
- Cetylpyridinium Chloride (0123-03-05)
- Climbazole (38083-17-9) (C15H17ClN2O2)
- Coco Monoethanolamide (CH3(CH2)nCONHCH2CH2OH)
- Coconut Oil (8001-31-8)
- Decyl Alcohol (112-30-1)
- Decyl Glucoside (58846-77-8)
- Diethyl Phthalate (84-66-2)
- Dihydroxyacetone (96-26-4) (C3H6O3)
- Dimethyl Isosorbide (5306-85-4)
- Dodecyl Benzene (C18H30)
- Dodecyl Sulfate Sodium Salt
- Dodecylbenzene Sulphonic Acid
- Dodecylbenzenesulfonic acid (27176-87-0)
- Ethylhexyl Oleate (26399-02-0)
- Hydroxypropyl Methyl Cellulose
- Glycerin (56-81-5) (C3H8O3)
- Glyceryl Monolaurate (27215-38-9)
- Glycolic Acid (79-14-1)
- Hydrogenated Palm Stearin
- Hydroxyethyl Cellulose (9004-62-0)
- Isododecane (31807-55-3) (C12H26)
- Isopropyl Myristate (110-27-0)
- Isothiazolinone (1003-07-02) (C3H3NOS)
- Kojic Acid (501-30-4)
- L-Ascorbyl Palmitate (137-66-6)
- Aminoethyl Ethanolamine (111-41-1)
- Lactic Acid 90%
- Lauric Acid (0143-07-07)
- Lauric Acid Diethanolamide
- Linear Alkyl Benzene Sulfonic Acid
- Linoleic Acid (60-33-3)
- Lutensol XL 80
- Magnesium Aluminium Silicate
- Methyl Hydrogenated Rosinate
- Microcrystalline Wax (63231-60-7)
- Mineral Oil (8042-47-5)
- Myristyl Alcohol (112-72-1)
- Octyl Stearate (109-36-4)
- Palmitic Acid (57-10-3)
- Paraffin Oil (8012-95-1)
- Peracetic Acid (79-21-0)
- Polyquaternium 47 (197969-51-0)
- Polysorbate 20 (9005-64-5)
- Potassium Oleate (143-18-0)
- Propylene Glycol Monostearate
- Rice Bran Wax Pastilles
- Soap Noodles (Sodium carboxylates)
- Sodium Dichloroisocyanurate (C3Cl2N3NaO3)
- Sodium Lauryl Ether Sulphate
- Sodium levulinate (19856-23-6)
- Sodium Metasilicate (6834-92-0)
- Sodium Monofluorophosphate (10163-15-2)
- Sodium Oleate (143-19-1)
- Sodium Picramate (831-52-7)
- Sodium Stearate (822-16-2)
- Sodium Tripolyphosphate (7758-29-4)
- Sodium xylenesulfonate (1300-72-7)
- Sorbitan Monooleate (1338-43-8)
- Sorbitan Monostearate (1338-41-6)
- Sorbitan Trioleate (26266-58-0)
- Sorbitol (50-70-4) (C6H14O6)
- Soya Wax (Soy wax)
- Spilanthol 20% (C14H23NO)
- Dodecyl Amine (124-22-1)
- Stearic Acid (57-11-4)
- Stearyl Alcohol (112-92-5)
- Talcum (14807-96-6) (H2Mg3O12Si4)
- Tetraacetylethylenediamine (10543-57-4) (C10H16N2O4)
- Thioglycolic Acid (68-11-1)
- Titanium Dioxide (13463-67-7)
- Titanium Dioxide Rutile 902
- Tributyl Citrate (77-94-1)
- Triethanolamine 99% (C6H15NO3)
- Trimethylsiloxysilicate (56275-01-05) (C3H12O4Si2)
- Trisodium Phosphate Anhydrous
- Triton X-100 (C16H26O2)
- Urea Hydrogen Peroxide
- Xanthan Gum (11138-66-2)
- Zeolite 4A (Powder)
- Zinc Ricinoleate (13040-19-2)
- 2-Ethylhexyl 4-Aminobenzoate (26218-04-02)
- 3-[(2-Ethylhexyl)Oxy]-1,2-Propanediol (70445-33-9) (C11H24O3)
- Aloe Vera (Aloe extract)
- Ammonium Lauryl Sulfate
- Beta-Carotene (7235-40-7) (C40H56)
- Caprylic/Capric Triglyceride (65381-09-01)
- Carnauba Wax (600 g/mol)
- Ceresin Wax (442.72 g/mol)
- Cetostearyl Alcohol (67762-27-0)
- Cetrimonium Chloride (0112-02-07)
- Chloroxylenol (88-04-0) (C8H9ClO)
- Cocamide DEA (68603-42-9)
- Cocamidopropyl Betaine (61789-40-0)
- Coconut Fatty Acid
- D-Panthenol (81-13-0) (C9H19NO4)
- Didecyldimethylammonium Chloride (7173-51-5)
- Dipropylene Glycol Monomethyl Ether
- Disodium Lauryl Sulfosuccinate
- Hydrogenated Palm Oil
- Iodophor (Liquid) (25000 g/mol)
- Macrogolglycerol Ricinoleate (61791-12-6)
- Niacinamide Ascorbate (1987-71-9)
- Nonylphenol Ethoxylate 9.5 Mole
- Octyl Methoxycinnamate (5466-77-3)
- Sodium Cocoyl Isethionate
- Sodium Dl-Lactate (72-17-3)
- Sodium Dodecylbenzenesulfonate (25155-30-0)
- Sodium Lauroyl Sarcosinate
- Sodium Methylparaben (5026-62-0)
- Sodium Perborate Anhydrous
- Sodium Perborate Tetrahydrate
- Sodium Propylparaben (35285-69-9)
- Sorbic Acid (110-44-1)
- Dowfax 2A1 (12626-49-2)
- Sorbitan Tristearate (26658-19-5)
- Triclosan (3380-34-5) (C12H7Cl3O2)
- Tween 20 (Polyoxyethylene sorbitan monolaurate)
- TWEEN 60 (C35H68O10)
- Tween 80 (Polyoxy ethylenesorbitan monooleate)
- 1,3-Butanediol (107-88-0) (C4H10O2)
- Aluminum Chlorohydrate Dihydrate
- Hyaluronic Acid (9004-61-9)
- N-Acetyl-D-Glucosamine (7512-17-6) (C8H15NO6)
- Baking Powder (Leavening Agent)
- Disodium Laureth Sulfosuccinate
- Fatty Alcohol Ethoxylate
- Glycerol Alpha-Monostearate
- Hydrogenated Soybean Oil
- Peanut Oil (8002-03-07)
- Petroleum Jelly (8009-03-08)
- Polyquaternium-7 (26590-05-6) (Liquid)
- Silicone Emulsion
- Sodium Bromate (7789-38-0)
- Sodium Chlorite 80%
- Xylitol (87-99-0) (C5H12O5)
- Zinc disodium EDTA 15%
- P-Phenylenediamine (106-50-3) (C6H8N2)
- Alcohol Ethoxylate C12-16
- Alpha Arbutin (84380-01-08)
- Cetylpyridinium Chloride monohydrate
- Dimethylaminopropylamine (109-55-7) (C5H14N2)
- Ethylparaben (120-47-8) (C9H10O3)
- Magnesium Ascorbyl Phosphate
- Sodium Phosphate Tribasic Dodecahydrate
- Glycerol Monooleate (0111-03-05)
- Sodium Orthosilicate (13472-30-5)
- Sodium Hypochlorite 5%
- C12-C14 Alcohol ethoxylate 8 Mole Emulsifier
- Stannous Fluoride (7783-47-3)
- Sodium Hypochlorite (7681-52-9)
- Vitamin E Acetate
- Calcium Thioglycolate Trihydrate
- Octyldodecanol (5333-42-6) (C20H42O)
- PEG-40 Hydrogenated Castor Oil
- 1,2 Hexanediol (6920-22-5) (C6H14O2)
- 2-Methylresorcinol (608-25-3) (C7H8O2)
- 4-Butylresorcinol (18979-61-8) (C10H14O2)
- Butyl Stearate (123-95-5)
- Piroctone Olamine (68890-66-4)
- Caprylic/Capric Acid (C10H20O2)
- Octocrylene (6197-30-4) (C24H27NO2)
- Polyethylene Glycol 8000
- Sodium Perborate Monohydrate
- Terephthalylidene Dicamphor Sulfonic Acid
- Tinogard TL (C25H35N3O)
- MCT Oil (Liquid)
- Trisodium Citrate Dihydrate
- Cationic Guar Gum
- Cottonseed Oil (8001-29-4)
- Glycerine CP (C3H8O3)
- Isotridecanol (27458-92-0) (C13H28O)
- Homosalate (118-56-9) (C16H22O3)
- Lauryl Myristyl Alcohol
- Dioctadecyl Dimethyl Ammonium Chloride
- Linear Alkyl Benzene Sulfonic Acid 96%
- Apricot Oil (Liquid)
- Argan Oil (Liquid)
- Camellia Sinensis Leaf Extract
- Capryloyl Glycine (14246-53-8)
- Coco Betaine (Liquid)
- Cyclopentasiloxane (0541-02-06) (C10H30O5Si5)
- Decyltrimethylammonium Bromide (2082-84-0)
- Dihydrogenated Tallow Dimethyl Ammonium Chloride
- Disodium EDTA Anhydrous
- Distilled Coconut Fatty Acid
- Dodecyl Pyridinium Chloride
- Dodecyltrimethylammonium Bromide (1119-94-4)
- Ethylene Glycol Monostearate
- Grape Seed Oil
- Hazelnut Oil (885.0 g/mol)
- Sodium Lauryl Ether Sulfate
- Myristyl Gamma Picolinium Chloride
- Sodium Lauryl Sulfate Powder
- Oleyl-Cetyl Alcohol Ethoxylate
- Sodium Metasilicate Pentahydrate
- Sodium Methyl Cocoyl Taurate
- Sodium Thioglycolate (367-51-1)
- P-Chloro-M-Cresol (59-50-7) (C7H7ClO)
- Sorbitan Monolaurate (1338-39-2)
- Soy Wax Flakes
- Iodopropynyl Butylcarbamate (55406-53-6)
- Trisiloxane Ethoxylate (720.0 g/mol)
- Undecylenic Acid (112-38-9)
- Jojoba Oil (Liquid)
- Wheat Germ Oil
- L-Lactic Acid (79-33-4)
- Ferulic Acid (1135-24-6)
- Lauramine Oxide (1643-20-5)
- Lauryl Alcohol Ethoxylate
- Light Liquid Paraffin Oil
- Macadamia Oil (438545-25-6)
- Palm Fatty Acid
- 4-Hydroxyacetophenone (99-93-4) (C8H8O2)
- Palm Fatty Acid Distillate
- EDTA Tetrasodium Tetrahydrate
- Polyquaternium-11 (53633-54-8) (Liquid)
- Sodium P-Toluenesulfonate (657-84-1)
- Sodium Lauryl Ether Sulphate 28% Solution
- Sodium Hypochlorite Pentahydrate
- Sodium Hydroxymethyl Glycinate
- Oregano Oil (liquid)
- Sodium Hyaluronate (9067-32-7)
- Sea Buckthorn Oil
- Witch Hazel Extract
- Glyoxyloyl Keratin Amino Acid
- Selenium Disulfide (7488-56-4)
- Sodium Dodecylbenzenesulfonate Powder
- Shea Butter (Solid)
- Dimethyloldimethyl Hydantoin (6440-58-0)
- Sodium Lactate 60% Solution
- Sodium Copper Chlorophyllin
- Macadamia Oil (438545-25-6)
- Sodium Glycinate (6000-44-8)
- PEG-7 Glyceryl Cocoate
- Peg-75 Lanolin (LIquid)
- 1,2-Pentanediol (5343-92-0) (C5H12O2)
- Polyquaternium-51 (Powder) (150000 g/mol)
- Bakuchiol (10309-37-2) (S-Bakuchiol)
- Polysorbate 85 (Liquid)
- Sesame Oil (Liquid)
- Bemotrizinol (187393-00-6) (C38H49N3O5)
- Sodium Lauryl Ether Sulfate 70%
- C12-15 Alkyl Benzoate
- Triclocarban (101-20-2) (C13H9Cl3N2O)
- Cetalkonium Chloride (122-18-9)
- Decyl Oleate (3687-46-5)
- Diethylamino Hydroxybenzoyl Hexyl Benzoate
- Dimethyl Lauramine (112-18-5)
- Sulfonic Acid (C6H7NO3S)
- Ensulizole (27503-81-7) (C13H10N2O3S)
- Ethylhexyl Stearate (22047-49-0)
- Avobenzone (70356-09-01) (C20H22O3)
- Glyceryl Stearate (C21H42O4)
- Laureth-4 (362.54 g/mol) (315 C)
- Acid Thickener (Oleyl Amine Ethoxylate)
- Peg-100 Stearate (Solid)
- Sodium Xylenesulfonate Liquid
- Methylisothiazolinone (2682-20-4) (C4H5NOS)
- Coco Amido Propyl Betaine
- Propylene Glycol Dioleate
- Ethylhexyl Palmitate (29806-73-3)
- Soya Lecithin Powder
- Eutanol G (118.17 g/mol)
- Alpha-Olefin Sulfonate (420 g/mol)
- PEG - 200 (Polyethylene Glycol 200)
- Palmitic Acid 95%
- Sunflower Fatty Acid
- Rice Bran Fatty Acid
- Refined Glycerin (C3H8O3)
- Glycerol Monostearate (123-94-4)
- Glyceryl Oleate (25496-72-4)
- PEG - 1500 (Polyethylene Glycol 1500)
- 4-Isopropyl-3-Methylphenol (02-02-3228) (C10H14O)
- Stearin (555-43-1) (Solid)
- Sorbitan Isostearate (54392-26-6)
- PEG-8 Distearate (Solid) (903.3 g/mol)
- PEG-15 Stearate (Polyethylene glycol 15 stearate) (944.0 g/mol)
- PEG-32 Distearate (Solid) (1942 g/mol)
- PEG-40 Stearate (Solid) (2046 g/mol)
- PEG-150 Stearate (Solid) (6892 g/mol)
- PEG-175 Distearate (Solid) (707.92 g/mol)
- PEG-600 Dioleate (Solid) (1129.0 g/mol)
- PEG-600 Monostearate (866.5 g/mol)
- Glyceryl Dilaurate (27638-00-2)
- Ceto Stearyl Alcohol 14
- Ceto Stearyl Alcohol 12
- Cetostearyl Alcohol 20
- Stearyl Alcohol Ethoxylate 21
- Ceto Stearyl Alcohol Ethoxylate 21
- Triceteareth-4 Phosphate (119415-05-3) (1360 g/mol)
- Ceteth-10 Phosphate (PEG-10 Cetyl Ether Phosphate)
- C 8 alcohol carboxylate 8
- C9-C11 Alcohol Ethoxylate 5
- Castor Oil Ethoxylate 25
- Hydrogenated Castor Oil Ethoxylate 40
- Glycerine Ethoxylate - 26
- Glycerine Ethoxylate - 27
- Oleyl alcohol carboxylate 9
- 1-Bromotetradecane (112-71-0) (Myristyl Bromide)
- Tocopherol (1406-66-2) (430.70 g/mol)
- Sodium Palmitate (408-35-5)
- PEG-80 Sorbitan Laurate
- Chlorphenesin (104-29-0) (Solid)
- 1,2-Decanediol (1119-86-4) (Liquid)
- Lauryl Alcohol 7 EO
- Butanol 13 EO
- Sorbitan Sesquioleate (8007-43-0)
- PPG-2 Myristyl Propionate (74775-06-07)
- PEG-200 Mono Laurate
- PEG-200 Di Laurate
- PEG-250 Monooleate (514.7 g/mol)
- PEG-400 Dioleate (929 g/mol)
- PEG 400 Dilaurate
- PEG-400 Mono Laurate
- PEG 600 Monooleate
- Glyceryl Monostearate (31566-31-1)
- C9-C11 Alcohol Ethoxylate 10
- Polysorbate 60 (Liquid)
- Polysorbate 65 (1310 g/mol)
- PEG-200 (Liquid) (198.22 g/mol)
- PEG-400 (Liquid) (400.40 g/mol)
- Sodium Dichloroisocyanurate Anhydrous
- Hydrogenated Castor Oil Ethoxylates
- Agrochemicals
- Nitric Acid (7697-37-2)
- 2,4-Dichlorophenol (120-83-2) (C6H4Cl2O)
- 2-Cyanopyridine (100-70-9) (C6H4N2)
- 2,5-Dichloroaniline (95-82-9) (C6H5Cl2N)
- M-Toluic Acid (99-04-7)
- Cyanuric Chloride (108-77-0)
- Isopropylamine (75-31-0) (C3H9N)
- Methyl 2-Chloropropionate (17639-93-9)
- Pyridine (110-86-1) (C5H5N)
- Chloroacetyl Chloride (79-04-9)
- Methyl Chloroacetate (96-34-4)
- Propylamine (0107-10-8) (C3H9N)
- Diisopropylamine (108-18-9) (C6H15N)
- Zinc Sulphate Anhydrous
- 2-Chlorobenzonitrile (873-32-5) (C7H4ClN)
- Cuelure (3572-06-03) (C12H14O3)
- Sodium Nitrate (7631-99-4)
- Azadirachtin (11141-17-6) (C35H44O16)
- Pyrethrum (8003-34-7) (C43H56O8)
- Potassium Sulphate (7778-80-5)
- Potassium Silicate (1312-76-1)
- Potassium Nitrate (7757-79-1)
- Magnesium Sulphate Anhydrous
- Ammonium Sulfamate (7773-06-0)
- Diatomaceous Earth (61790-53-2)
- 1,2-Dibromoethane (106-93-4) (C2H4Br2)
- Zinc Sulphate Heptahydrate
- 2,6-Difluorobenzamide (18063-03-01) (C7H5F2NO)
- 1,2,4-Triazole (288-88-0) (C2H3N3)
- Bromadiolone (28772-56-7) (C30H23BrO4)
- 2,6-Dichlorobenzonitrile (1194-65-6) (C7H3Cl2N)
- Neem Oil (875 g/mol)
- Calcium Cyanamide (156-62-7)
- Liquor Ammonia (NH4OH)
- Organic Manure (180.0 g/mol)
- Calcium Nitrate (10124-37-5)
- Calcium Peroxide (78403-22-2)
- Monoammonium Phosphate (NH4H2PO4)
- Acetoacet Ortho Chloroanilide
- Rock Phosphate (504.30 g/mol)
- Cyanamide (0420-04-02) (CH2N2)
- Tributyltin Oxide (56-35-9)
- Copper Oxychloride (1332-40-7)
- Urea (57-13-6) (CH4N2O)
- Zinc Phosphide (1314-84-7)
- Triacontanol (593-50-0) (C30H62O)
- 1-Naphthaleneacetic Acid (86-87-3)
- Temephos (3383-96-8) (C16H20O6P2S3)
- Gibberellic Acid (77-06-5)
- Sulfur (7704-34-9) (S)
- Sodium Pentachlorophenate (131-52-2)
- Sodium Humate (68131-04-04)
- Potassium Humate (68514-28-3)
- Potassium Diformate (C2H3KO4)
- Fulvic Acid (479-66-3)
- Humic Acid (1415-93-6)
- Disodium Octaborate Tetrahydrate
- Cyromazine (66215-27-8) (C6H10N6)
- Custard Apple Seed Extract
- Buta phosphan (C7H18NO2P)
- 2,4,6-Trichlorophenol (88-06-2) (C6H3Cl3O)
- Propoxur (114-26-1) (C11H15NO3)
- Manganese Sulphate Anhydrous
- Manganese Carbonate (598-62-9)
- Paraquat Dichloride (1910-42-5)
- Malathion (121-75-5) (C10H19O6PS2)
- Magnesium Nitrate Hexahydrate
- Nicotine Sulphate (65-30-5)
- Mono Potassium Phosphate
- Hexaconazole (79983-71-4) (C14H17Cl2N3O)
- Ammonium Sulfate (7783-20-2)
- Ammonia (7664-41-7) (NH3)
- Amitraz (33089-61-1) (C19H23N3)
- Ammonium Dihydrogen Phosphate
- Disodium Octaborate Anhydrous
- Dicyandiamide (461-58-5) (C2H4N4)
- EDTA Ferric Sodium
- 1,2,4-Triazole Sodium Salt
- Ammonium Lignosulfonate (8061-53-8)
- Tallow Amine Ethoxylate
- Monomethylamine (74-89-5) (CH3NH2)
- Diammonium Phosphate (7783-28-0)
- 2,6-Dichlorobenzaldehyde (83-38-5) (C7H4Cl2O)
- Phosphoric Acid (7664-38-2)
- Tricalcium phosphate (7758-87-4)
- Potassium Carbonate (0584-08-07)
- Potassium Chloride (7447-40-7)
- Chlormequat Chloride (999-81-5)
- Magnesium Sulphate Heptahydrate
- Imidacloprid (138261-41-3) (C9H10ClN5O2)
- Lambda-Cyhalothrin (91465-08-06) (C23H19ClF3NO3)
- Cypermethrin (52315-07-08) (C22H19Cl2NO3)
- Cloquintocet-Mexyl (99607-70-2) (C18H22ClNO3)
- Magnesium Sulfate Monohydrate
- Karanja Oil (247588-54-1)
- 2,4-Dichlorophenoxyacetic Acid (94-75-7)
- 3-Phenoxybenzaldehyde (39515-51-0) (C13H10O2)
- Thiophanate Methyl (23564-05-08)
- Sulphur (S) (32.065 g/mol)
- 6-Benzylaminopurine (1214-39-7) (C12H11N5)
- Deltamethrin (52918-63-5) (C22H19Br2NO3)
- Permethrin (52645-53-1) (C21H20Cl2O3)
- Copper Sulfate Pentahydrate
- 1,4-Dichlorobenzene (106-46-7) (C6H4Cl2)
- Manganese Sulphate Monohydrate
- Sulfur Powder (S)
- Diethyl Aminoethyl Hexanoate
- Cyanamide Solution 50%
- Indole-3-Butyric Acid (133-32-4)
- Indole-3-Acetic Acid (87-51-4)
- Humic Acid Powder
- Humic Acid Granules
- Tebuconazole (107534-96-3) (C16H22ClN3O)
- Tricyclazole (41814-78-2) (C9H7N3S)
- 3,5-Dichlorobenzoyl Chloride (2905-62-6)
- Metribuzin (21087-64-9) (C8H14N4OS)
- Thiamethoxam (153719-23-4) (C8H10ClN5O3S)
- Theta-Cypermethrin (71697-59-1) (C22H19Cl2NO3)
- Ethion (0563-12-2) (C9H22O4P2S4)
- Alpha-Cypermethrin (67375-30-8) (C22H19Cl2NO3)
- Cypermethric Acid Chloride
- Gibberellic Acid 0.186% SP
- Ferrous Sulphate 19%
- Lindane (58-89-9) (C6H6Cl6)
- 3-Phenoxybenzyl Alcohol (13826-35-2)
- Monocalcium Phosphate Feed Grade
- Metamidophos (10265-92-6) (C2H8NO2PS)
- Spinosad (168316-95-8) (C83H132N2O20)
- Sodium Selenate (13410-01-0)
- 2,3,5,6-Tetrachloro Pyridine (C5HCl4N)
- Ferrous Sulphate Monohydrate
- 2,3-Dichloronitrobenzene (3209-22-1) (C6H3Cl2NO2)
- 3,5-Dimethylpyrazole (67-51-6) (C5H8N2)
- Chloral Anhydrous (75-87-6)
- Potassium Thiosulfate (10294-66-3)
- 4-Acetylmorpholine (1696-20-4) (C6H11NO2)
- Spent Sulphuric Acid
- Potassium Silicate Liquid
- Zinc EDTA (C10H12N2O8Zn2)
- Sulfur Granules (Solid)
- 1,2-Dichlorobenzene (95-50-1) (C6H4Cl2)
- Sulfur Lumps (S)
- Chlorpyrifos 20% (Chlorpyrifos 20% EC)
- Glyphosate 41% (Liquid)
- Dimethoate 30% (Liquid)
- Superphosphate (8011-76-5) (Single Superphosphate)
- Profenofos (Liquid) (328.13 g/mol)
- Diethyl Phenyl Acetamide
- 1,2-Dibromo-3-Chloropropane (96-12-8) (C3H5Br2Cl)
- Nonyl Phenol Ethoxylate
- Plastic & Rubber Chemicals
- Zinc Stearate (0557-05-01)
- Acetone Cyanohydrin (75-86-5)
- Calcium Stearate (1592-23-0)
- Alpha-Methylstyrene (98-83-9) (C9H10)
- Decabromodiphenyl Ethane (84852-53-9)
- Zinc Oxide (1314-13-2)
- Zinc Hydroxide (20427-58-1)
- Triphenyl Phosphite (101-02-0)
- Triphenyl Phosphate (115-86-6)
- Ammonium Polyphosphate (68333-79-9)
- Ammonium Stearate (1002-89-7)
- Tricresyl Phosphate (1330-78-5)
- Toluene Diisocyanate (26471-62-5)
- Tetrabromobisphenol A (79-94-7)
- 1,4-Butanediol (110-63-4) (1,4-Dihydroxybutane)
- 2-Acrylamido-2-Methylpropane Sulfonic Acid
- 3-Glycidyloxypropyltrimethoxysilane (2530-83-8) (C9H20O5Si)
- Polyester Resins (Liquid)
- Triallyl Isocyanurate (1025-15-6)
- Triallyl Cyanurate (101-37-1)
- Cellulose Acetate (9004-35-7)
- Pigment Red 4
- Cobalt Octoate (136-52-7)
- Bisphenol A (80-05-7)
- 2-Ethylhexyl Acrylate (0103-11-7)
- 2-Hydroxyethyl Acrylate (818-61-1)
- Polyethylene Wax (Solid)
- Caprolactam (105-60-2) (C6H11NO)
- 2-Mercaptobenzothiazole (149-30-4) (C7H5NS2)
- Ketonic Resin (Solid)
- Polyamide resin (Solid)
- Cellulose Acetate Butyrate
- Divinylbenzene (1321-74-0) (C10H10)
- Polytetramethylene Ether Glycol
- Phenol Formaldehyde Resin
- N-Tert-Butylacrylamide (107-58-4) (C7H13NO)
- Epoxy Resin (55818-57-0)
- Montan Wax (750 g/mol)
- Chlorinated Polyethylene (64754-90-1)
- Diphenylguanidine (0102-06-07) (C13H13N3)
- Hydroxypropyl Acrylate (25584-83-2)
- Acrylamide (79-06-1) (C3H5NO)
- Azodicarbonamide (123-77-3) (C2H4N4O2)
- Polypropylene (9003-07-0) ([CH2CH(CH3)]n)
- N-Acetylcaprolactam (1888-91-1) (C8H13NO2)
- Aluminium Stearate (0637-12-7)
- Benzoguanamine (91-76-9) (C9H9N5)
- Benzothiazole (95-16-9) (C7H5NS)
- Trimellitic Anhydride
- Trimellitic Anhydride (552-30-7)
- Polylactic Acid (26100-51-6)
- Redispersible Polymer (1000000 g/mol)
- Dodecanedioic Acid (693-23-2)
- Dimethyl Terephthalate (120-61-6)
- Diacetone Acrylamide (2873-97-4)
- Dicyclopentadiene (77-73-6) (C10H12)
- Phthalic Anhydride (85-44-9)
- P-Toluenesulfonamide (70-55-3) (C7H9NO2S)
- N,N'-Methylenebisacrylamide (110-26-9) (C7H10N2O2)
- O-Toluenesulfonamide (88-19-7) (C7H9NO2S)
- Trimethylolpropane (77-99-6) (C6H14O3)
- Triethylenediamine (280-57-9) (C6H12N2)
- Tert-Butyl Peroxybenzoate (614-45-9)
- Methyl Ethyl Ketone Peroxide
- Strontium Hydroxide (18480-07-04)
- Sodium Polyacrylate (9003-04-07)
- Benzyl Butyl Phthalate
- Para-Tertiary Butylphenol (98-54-4)
- Polyvinyl Chloride (9002-86-2)
- Polyvinyl Butyral (63148-65-2)
- Polyvinyl Acetate (9003-20-7)
- Polyurethane (Solid) (100000 g/mol)
- Polymethyl Methacrylate (9011-14-7)
- Lead Stearate (1072-35-1)
- Ethylene Vinyl Acetate
- Glycidyl Methacrylate (106-91-2)
- Ethylene Bisstearamide (C38H76N2O2)
- Dipropylene Glycol Dibenzoate
- Erucamide (112-84-5) (C22H43NO)
- Dipentaerythritol (126-58-9) (C10H22O7)
- Dioctyl Adipate (123-79-5)
- Dimethyl Adipate (627-93-0)
- Dimer Acid (61788-89-4)
- Isophthalic Acid (121-91-5)
- Diethylene Glycol Dibenzoate
- Dicumyl Peroxide (80-43-3)
- Dibutyltin Dilaurate (77-58-7)
- Chlorinated Paraffin Wax
- Cyclohexanone Formaldehyde Resin
- Antimony Trioxide (Sb2O3)
- Itaconic Acid (97-65-4)
- Tin 2-Ethylhexanoate (301-10-0)
- Polyethylene Glycol Diacrylate
- Retarder (C13H10Cl3NO2S2) (218.14 g/mol)
- Di-Tert-Butyl Peroxide (0110-05-04)
- Benzylamine (100-46-9) (C7H9N)
- Dodecyl Mercaptan (112-55-0)
- Dioctyl Phthalate (117-81-7)
- Dioctyl Maleate (2915-53-9)
- Diallyl Phthalate (131-17-9)
- Diallyl Maleate (999-21-3)
- Diisononyl Phthalate (28553-12-0)
- Dicyclohexyl Phthalate (84-61-7)
- Dibutyl Sebacate (109-43-3)
- Dibutyl Phthalate (84-74-2)
- Dibutyl Maleate (105-76-0)
- Dibutyl Adipate (105-99-7)
- Ammonium Polyacrylate (9003-03-06)
- 3-Aminopropyltrimethoxysilane (13822-56-5) (C6H17NO3Si)
- 3-Aminopropyltriethoxysilane (919-30-2) (C9H23NO3Si)
- Aluminium Trihydrate (Al(OH)3.3H2O)
- Acrylonitrile (107-13-1) (C3H3N)
- Propylene Oxide (75-56-9)
- 3-Trimethoxysilyl propyl methacrylate
- Methyl Methacrylate (80-62-6)
- Polyethylene (9002-88-4) ((C2H4)n)
- Bis-2-Propylheptyl Phthalate (53306-54-0)
- Urea Formaldehyde (Solid)
- Polybutylene Succinate (25777-14-4)
- Polydimethylsiloxane Vinyl Terminated
- Silicone Resin (organopolysiloxane resin)
- Allyl Glycidyl Ether
- 3-Mercaptopropyl Trimethoxysilane (4420-74-0)
- Acrylic Emulsion (150000 g/mol)
- Acrylic Thickener (1000000 g/mol)
- PBR CISAMER 01
- Polypropylene Glycol (25322-69-4)
- Barium Stearate (6865-35-6)
- Allyl Methacrylate (96-05-9)
- Hydroxyethyl Methacrylate (868-77-9)
- Diisodecyl Adipate (27178-16-1)
- Butyl Acrylate (141-32-2)
- 4-Tert-Octylphenol (140-66-9) (C14H22O)
- Vinyl Acetate Monomer
- Trimethylolpropane Triacrylate (15625-89-5)
- Hexamethylenediamine (0124-09-04) (C6H16N2)
- Methyl Acrylate (96-33-3)
- Methacrylic Acid (79-41-4)
- Methacrylamide (79-39-0) (C4H7NO)
- Melamine Cyanurate (37640-57-6)
- Melamine (108-78-1) (C3H6N6)
- Pentaerythritol (115-77-5) (C5H12O4)
- Oleamide (301-02-0) (C18H35NO)
- Neopentyl Glycol (126-30-7)
- N-Octyl Mercaptan (111-88-6)
- Phthalic Acid (88-99-3)
- 2-Ethylhexanol (104-76-7) (C8H18O)
- Acetyl Tributyl Citrate
- 4-tert-Butylbenzoic acid (98-73-7)
- Ammonium Persulphate (7727-54-0)
- Hydroxylammonium Sulfate (10039-54-0)
- Chrome Alum (7788-99-0)
- Ethylene Diamine Acid Phosphate
- Tetrachlorophthalic Anhydride (0117-08-08)
- Terephthalic Acid (100-21-0)
- Benzoyl Chloride (98-88-4)
- Monoethylene Glycol (107-21-1)
- Cyclohexane (110-82-7) (C6H12)
- Dibutylamine (111-92-2) (C8H19N)
- Methyl Vinyl Ketone
- Dimethyl Phthalate (0131-11-3)
- Epichlorohydrin (106-89-8) (C3H5ClO)
- Ethyl vinyl ether
- Tetraethylene Pentamine (112-57-2)
- Propionic Anhydride (123-62-6)
- Amorphous Silica (SiO2)
- Zinc Borate Anhydrous
- Diphenyl Isodecyl Phosphate
- Ethylene (74-85-1) (C2H4)
- PVC Resin K67
- Polyacrylic Acid (9003-01-04)
- P-Xylene (106-42-3) (C8H10)
- Phenyl Isocyanate (103-71-9)
- Acetyl Acetone Peroxide
- Zinc Borate (B2O6Zn3)
- Bis-2-Ethylhexyl Sebacate (122-62-3)
- Sulphur Monochloride (10025-67-9)
- Maleic Acid (110-16-7)
- Phenol (108-95-2) (C6H6O)
- Phosphorous Acid (13598-36-2)
- 4-Tert-Butylcatechol (98-29-3) (C10H14O2)
- Acrylic Acid (79-10-7)
- Acrylic Resin (100000 g/mol)
- Adipic Acid (0124-04-09)
- Hexamethylenetetramine (100-97-0) (C6H12N4)
- Maleic Anhydride (108-31-6)
- Paraformaldehyde (30525-89-4) (HO(CH2O)nH)
- 1,2-Dichloroethane (0107-06-02) (C2H4Cl2)
- Diethylenetriamine (111-40-0) (C4H13N3)
- Erucic Acid (112-86-7)
- PMMA (Acrylic Resin) ((C5H8O2)n)
- Dioctyl Sebacate1 (2432-87-3)
- Dimethyl Sebacate (106-79-6)
- 1-Hydroxycyclohexyl Phenyl Ketone
- Polyvinyl Chloride SG5
- Polyalphaolefin (420 g/mol) (320 C)
- Methylene Diphenyl Diisocyanate
- Butyl Methacrylate (97-88-1)
- N-Vinylpyrrolidone (88-12-0) (C6H9NO)
- M-Xylylenediamine (1477-55-0) (C8H12N2)
- Isobutyl Methacrylate (97-86-9)
- Ethylene Vinyl Alcohol
- Diisodecyl Phthalate (26761-40-0)
- 12-Hydroxy Stearic Acid
- Epoxidized Soyabean Oil
- 1,3-Propanediol (504-63-2) (C3H8O2)
- Aibn (78-67-1) (C8H12N4)
- Vinyltrimethoxysilane (07-02-2768) (C5H12O3Si)
- 2-Methyl-1,3-Propanediol (2163-42-0) (C4H10O2)
- Dicyclohexylamine (101-83-7) ((C6H11)2NH)
- Melamine Powder (126.12 g/mol)
- Tripropylene Glycol Diacrylate
- Polyurethane Resin (50000 g/mol)
- Ethyl Methacrylate (97-63-2)
- Polybutene (9003-29-6) (60000 g/mol)
- Methacryloyl Chloride (920-46-7)
- Bis-3-Triethoxysilyl Propyl Tetrasulphide
- Dodecenylsuccinic Anhydride (25377-73-5)
- Stearyl Acrylate (4813-57-4)
- Polyaniline (25233-30-1) (50000 g/mol)
- Plioflex (9003-55-8) (250 g/mol)
- Isophorone Diisocyanate (4098-71-9)
- Dipropylene Glycol Diacrylate
- Dimethylaminoethyl Methacrylate (2867-47-2)
- 1,6-Hexanediol Diacrylate (13048-33-4)
- 1,6-Hexanediol (0629-11-8) (C6H14O2)
- 1-Octene (111-66-0) (C8H16)
- 3,5-Diaminobenzoic Acid (535-87-5)
- 4,4-Dihydroxydiphenyl Sulphone (80-09-1)
- PVC Resin K 67
- Residue Wax (310.50 g/mol)
- Jeffamine D-230 Polyetheramine
- Cardanol (37330-39-5) (C21H30O)
- Polymethylene Polyphenyl Polyisocyanate
- Novolac Phenolic Resin
- Polytetramethylene Ether Glycol-2000
- Epoxy Resin Ker 828
- Butyl Oleate (142-77-8)
- Vinyl Bromide (593-60-2)
- Carbon Black (1333-86-4)
- Tricyclodecanedimethanol (26896-48-0) (C12H20O2)
- Zinc Diethyldithiocarbamate (14324-55-1)
- Diisobutyl Phthalate (84-69-5)
- Alkyd Resin (Liquid)
- Polybutene 1300 ((C4H8)x)
- 2,2,4-Trimethyl-1,2-Dihydroquinoline (147-47-7) (C12H15N)
- Isophoronediamine (2855-13-2) (C10H22N2)
- Lotader 4210 (Solid)
- Cadmium Stearate (2223-93-0)
- 1,9-Decadiene (1647-16-1) (138.25 g/mol)
- Pharma Chemicals
- Ammonium Carbonate (506-87-6)
- Clonidine Related Compound A
- Alpha Picolinic Acid
- Aluminium Phosphate (7784-30-7)
- Aluminium Acetate (C6H9AlO6)
- Choline Bitartrate (87-67-2)
- Sodium Propionate (137-40-6)
- Magnesium Glycinate (14783-68-7)
- Sodium Salicylate (54-21-7)
- Sodium Silicofluoride (16893-85-9)
- Sodium Erythorbate (6381-77-7)
- Lactose Monohydrate (5989-81-1)
- Calcium Lactate (814-80-2)
- Polyethylene Glycol (25322-68-3)
- Magnesium Stearate (557-04-0)
- Guanidine Hydrochloride (50-01-1)
- Thioacetic Acid (C2H4OS)
- Sodium fluoride (7681-49-4)
- N-Methylphthalimide (550-44-7) (C9H7NO2)
- Calcium Acetate Anhydrous
- 3-Bromophenol (591-20-8) (C6H5BrO)
- 2-Aminopyridine (504-29-0) (C5H6N2)
- 4-Bromoaniline (106-40-1) (C6H6BrN)
- 4-Chlorobenzaldehyde (104-88-1) (C7H5ClO)
- 4-Nitrobenzaldehyde (555-16-8) (CaCl2)
- Ammonium Bicarbonate (1066-33-7)
- Ammonium Bromide (12124-97-9)
- Acetanilide (103-84-4) (C8H9NO)
- Alginic Acid (9005-32-7)
- Benzhydrol (91-01-0) (C13H12O)
- Malonic Acid (141-82-2)
- Pyridine-3-Sulfonyl Chloride (16133-25-8)
- Cis Bromo Benzoate
- Chlorhexidine (55-56-1) (C22H30Cl2N10)
- Dimethylamine Hydrochloride (506-59-2)
- Dipicolinic Acid (499-83-2)
- Gallic Acid Anhydrous
- Glutaric Anhydride (108-55-4)
- Potassium Bromate (KBrO3)
- Sodium Bisulphite (7631-90-5)
- Sodium Iodide Anhydrous
- Silver Nitrate (7761-88-8)
- Sodium Tartrate (C4H4Na2O6)
- Sucrose (57-50-1) (C12H22O11)
- Succinimide (123-56-8) (C4H5NO2)
- Tetraethylammonium Chloride (56-34-8)
- Triethylamine Hydrochloride (554-68-7)
- 3-Aminocrotononitrile (1118-61-2) (C4H6N2)
- Iron Sulfate Heptahydrate
- 4-Aminoveratrole (6315-89-5) (C8H11NO2)
- Sodium Acetate (C2H3NaO2)
- 2-Chloropropionyl Chloride (C3H4Cl2O)
- 3,5,5-Trimethylhexanoyl Chloride (36727-29-4)
- 4-Bromotoluene (106-38-7) (C7H7Br)
- Propylene Glycol (57-55-6)
- Isobutyl Bromide (78-77-3)
- Isobutyric Anhydride (97-72-3)
- Isobutylamine (78-81-9) (C4H11N)
- Para-Chlorobenzoyl Chloride (122-01-0)
- Methyl Chloroformate (79-22-1)
- 5-Chlorovaleroyl Chloride (1575-61-7)
- Dichloromethane (75-09-2) (CH2Cl2)
- Ethyl Bromoacetate (105-36-2)
- Ethyl Chloroacetate (105-39-5)
- Polysorbate 80 (9005-65-6)
- Methoxyacetyl Chloride (38870-89-2)
- Aspartame (22839-47-0) (C14H18N2O5)
- Dipotassium Phosphate Anhydrous
- Hydroxypropyl Cellulose (9004-64-2)
- Magnesium Phosphate (7757-87-1)
- Maltodextrin (9050-36-6) (C6nH(10n+2)O(5n+1))
- Polydextrose (68424-04-04) (2000 g/mol)
- Potassium Metabisulfite (16731-55-8)
- Potassium Phosphate (7778-53-2)
- Methyl Carbazate (6294-89-9)
- Calcium Propionate (4075-81-4)
- Cyclopropanecarbonyl Chloride (4023-34-1)
- Dextrose Anhydrous (50-99-7)
- Potassium Bicarbonate (298-14-6)
- L+Tartaric Acid (87-69-4)
- Ethylenediaminetetraacetic Acid Tripotassium Salt Dihydrate
- Sodium Carboxymethyl Cellulose
- Sodium Caseinate (9005-46-3)
- Sodium Selenite (10102-18-8)
- Edta Calcium Disodium
- Succinylcholine Chloride (71-27-2)
- Potassium Bisulfate (7646-93-7)
- Trisodium Citrate Anhydrous
- Glucose Syrup (180.16 g/mol)
- Allylamine (0107-11-9) (C3H8ClN)
- Carboxymethyl Cellulose (C8H16O8)
- 2-(2-Chloroethoxy)Ethanol (628-89-7) (C4H9ClO2)
- 3-Chloro-4-Fluoroaniline (367-21-5) (C6H5ClFN)
- Tert-Butylhydroquinone (1948-33-0) (C10H14O2)
- Thiourea (62-56-6) (CH4N2S)
- 4-Bromophenol (106-41-2) (C6H5BrO)
- Poloxamer 407 (C572H1146O259)
- Propionitrile (107-12-0) (C3H5N)
- Propionyl Chloride (79-03-8)
- 3-AcetylPyridine (0350-03-08) (C7H7NO)
- Calcium Formate (544-17-2)
- Disodium Phosphate Anhydrous
- DL-Tartaric Acid (133-37-9)
- Potassium Benzoate (582-25-2)
- Potassium Bromide (BrK)
- Sodium Benzoate (532-32-1)
- Sodium Bicarbonate (144-55-8)
- Sodium Ferrocyanide (13601-19-9)
- Sodium Metabisulphite (7681-57-4)
- Thiophenol (108-98-5) (C6H6S)
- Sodium Hydroxide Flakes
- Zinc Picolinate (17949-65-4)
- Zoledronic Acid (165800-06-6)
- Bismuth Subnitrate (1304-85-4)
- Bismuth Subsalicylate (14882-18-9)
- 2-Amino-5-Chlorobenzophenone (719-59-5) (C13H10ClNO)
- 2-Aminomethyl-1-Ethylpyrrolidine (26116-12-1) (C7H16N2)
- Busulfan (55-98-1) (C6H14O6S2)
- 2-Mercapto-5-Methoxybenzimidazole (37052-78-1) (C8H8N2OS)
- Caffeine (58-08-2) (C8H10N4O2)
- Caffeine Citrate (69-22-7)
- Calcium Bisglycinate (35947-07-0)
- Calcium D-Saccharate (5793-89-5)
- Calcium Gluconate (66905-23-5)
- Calcium Glycerophosphate (27214-00-2)
- 4-Aminobenzoic Acid (150-13-0)
- 4-Chlorosalicylic Acid (5106-98-9)
- Canagliflozin Hemihydrate (928672-86-0)
- Candesartan Cilexetil (145040-37-5)
- Carbamazepine (298-46-4) (C15H12N2O)
- Carbidopa (28860-95-9) (C10H14N2O4)
- Activated Charcoal (7440-44-0)
- Acyclovir (59277-89-3) (C8H11N5O3)
- Adapalene (106685-40-9) (C28H28O3)
- Carprofen (53716-49-7) (C15H12ClNO2)
- Carvedilol (72956-09-03) (C24H26N2O4)
- Celecoxib (169590-42-5) (C17H14F3N3O2S)
- Cellulose Acetate Phthalate
- Cetirizine Dihydrochloride (83881-52-1)
- Citicoline Sodium (33818-15-4)
- 5-Chlorosalicylic Acid (321-14-2)
- Clindamycin Phosphate (24729-96-2)
- Clomiphene Citrate (50-41-9)
- Clonidine Hydrochloride (4205-91-8)
- Clopidogrel Bisulfate (120202-66-6)
- 5-Sulphosalicylic Acid (97-05-2)
- Clotrimazole (23593-75-1) (C22H17ClN2)
- Abiraterone Acetate (154229-18-2)
- Amodiaquine Hydrochloride (6398-98-7)
- Ascomycin (104987-12-4) (C43H69NO12)
- Acetylsalicylic Acid (50-78-2)
- Acriflavine Hydrochloride (8063-24-9)
- Astaxanthin (472-61-7) (C40H52O4)
- Azithromycin Anhydrous (83905-01-05)
- Baclofen (1134-47-0) (C10H12ClNO2)
- Barbituric Acid (67-52-7)
- Albendazole (54965-21-8) (C12H15N3O2S)
- Benfotiamine (22457-89-2) (C19H23N4O6PS)
- Benzocaine (94-09-7) (C9H11NO2)
- Benzoic Acid (65-85-0)
- Indomethacin (53-86-1) (C19H16ClNO4)
- Inulin (9005-80-5) (5000 g/mol)
- Iodine (7553-56-2) (I2)
- Ipriflavone (35212-22-7) (C18H16O3)
- Beta Cyclodextrin (7585-39-9)
- Isonicotinamide (1453-82-3) (C6H6N2O)
- Betamethasone Dipropionate (5593-20-4)
- Biotin (58-85-5) (C10H16N2O3S)
- Docusate Sodium (0577-11-7)
- Itraconazole (84625-61-6) (C35H38Cl2N8O4)
- Ivermectin (70288-86-7) (875.10 g/mol)
- Ketoprofen (22071-15-4) (C16H14O3)
- L-Arginine Alpha-Ketoglutarate (16856-18-1)
- L-Arginine L-Aspartate (7675-83-4)
- l-Arginine Monohydrochloride (1119-34-2)
- L-Carnitine (541-15-1) (C7H15NO3)
- Hydrochlorothiazide (58-93-5) (C7H8ClN3O4S2)
- Hydrocortisone (50-23-7) (C21H30O5)
- L-Carnosine (305-84-0) (C9H14N4O3)
- Hydroxychloroquine Sulfate (747-36-4)
- L-Cysteine (52-90-4) (C3H7NO2S)
- Hydroxyethyl Starch (9005-27-0)
- L-Cystine (56-89-3) (C6H12N2O4S2)
- L-Glutamine (56-85-9) (C5H10N2O3)
- Ibuprofen (15687-27-1) (C13H18O2)
- L-Histidine (71-00-1) (C6H9N3O2)
- Hydroxyprogesterone Caproate (630-56-8)
- L-Leucine (61-90-5) (C6H13NO2)
- L-Mandelic Acid (17199-29-0)
- L-Methionine (63-68-3) (C5H11NO2S)
- L-Phenylalanine (63-91-2) (C9H11NO2)
- L-Pyroglutamic Acid (98-79-3)
- Montelukast Sodium (151767-02-1)
- Mupirocin (12650-69-0) (C26H44O9)
- L-Threonine (72-19-5) (C4H9NO3)
- L-Tryptophan (73-22-3) (C11H12N2O2)
- L-Valine (72-18-4) (C5H11NO2)
- N-Butyl Cyanoacrylate (6606-65-1)
- Lactitol Anhydrous (585-86-4)
- Lansoprazole (103577-45-3) (C16H14F3N3O2S)
- Lanthanum Carbonate (587-26-8)
- Nabumetone (42924-53-8) (C15H16O2)
- Alfuzosin Hydrochloride (81403-68-1)
- Alprostadil (745-65-3) (C20H34O5)
- L-Selenomethionine (3211-76-5) (C5H11NO2Se)
- N,N-Dicyclohexylcarbodiimide (538-75-0) (C13H22N2)
- Nalidixic Acid (0389-08-02)
- Naltrexone Hydrochloride (16676-29-2)
- Nebivolol (99200-09-06) (C22H25F2NO4)
- Leflunomide (75706-12-6) (C12H9F3N2O2)
- Lenalidomide (191732-72-6) (C13H13N3O3)
- Niacin (59-67-6) (C6H5NO2)
- Nicardipine Hydrochloride (54527-84-3)
- Letrozole (112809-51-5) (C17H11N5)
- Levodopa (59-92-7) (C9H11NO4)
- Levosulpiride (23672-07-03) (C15H23N3O4S)
- Lidocaine (137-58-6) (C14H22N2O)
- Niclosamide (50-65-7) (C13H8Cl2N2O4)
- Nicotinamide (98-92-0) (C6H6N2O)
- Nitazoxanide (55981-09-04) (C12H9N3O5S)
- Locust Bean Gum
- Loperamide Hydrochloride (34552-83-5)
- Lumefantrine (82186-77-4) (C30H32Cl3NO)
- Magnesium Acetyl Taurate
- Olmesartan Medoxomil (144689-63-4)
- Magnesium Citrate (3344-18-1)
- Ondansetron (99614-02-05) (C18H19N3O)
- Orlistat (96829-58-2) (C29H53NO5)
- Magnesium Hydroxide (1309-42-8)
- Orphenadrine Citrate (4682-36-4)
- Magnesium Lactate Anhydrous
- Oxybutynin Chloride (1508-65-2)
- Magnesium orotate (34717-03-08)
- Magnesium Pidolate (62003-27-4)
- Pantoprazole Sodium (138786-67-1)
- Para-Hydroxyphenylacetic Acid (156-38-7)
- Mandelic Acid (90-64-2)
- Manganese Gluconate (6485-39-8)
- Meclizine (569-65-3) (C25H27ClN2)
- Meglumine (6284-40-8) (C7H17NO5)
- Phenothiazine (92-84-2) (C12H9NS)
- Metformin Hydrochloride (1115-70-4)
- Phenylbutazone (50-33-9) (C19H20N2O2)
- Methylsulfonylmethane (67-71-0) (C2H6O2S)
- Metoclopramide (364-62-5) (C14H22ClN3O2)
- Pralidoxime Iodide (94-63-3)
- Pramoxine Hydrochloride (637-58-1)
- Pravastatin Sodium (81131-70-6)
- Miglustat (72599-27-0) (C10H21NO4)
- Pregabalin (148553-50-8) (C8H17NO2)
- Progesterone (57-83-0) (C21H30O2)
- Minoxidil (38304-91-5) (C9H15N5O)
- 4-Methoxy Phenylacetic Acid
- Pyridostigmine Bromide (101-26-8)
- Pyrimethamine (58-14-0) (C12H13ClN4)
- Alendronate Sodium (121268-17-5)
- 2-Phenylethylamine (64-04-0) (C8H11N)
- Nitrofurantoin (67-20-9) (C8H6N4O5)
- Oxiconazole Nitrate (64211-46-7)
- Methotrexate (59-05-2) (C20H22N8O5)
- Methylprednisolone Aceponate (86401-95-8)
- Amino Acid (110.09 g/mol)
- Chlortetracycline (478.45 g/mol) (380 Degree C)
- Chlorthalidone (77-36-1) (C14H11ClN2O4S)
- Choline Chloride (67-48-1)
- Choline Dihydrogen Citrate
- Choline Salicylate (2016-36-6)
- Bismuth Carbonate (597.98 g/mol)
- Chromium Picolinate (14639-25-9)
- Calcium Ascorbate (390.32 g/mol)
- Calcium Folinate (1492-18-8)
- Calcium Hydrogen Phosphate
- Copper Gluconate (0527-09-3)
- Calcium Phosphoryl Choline Chloride
- Croscarmellose Sodium (74811-65-7)
- Chloramphenicol (56-75-7) (C11H12Cl2N2O5)
- Chlorhexidine Gluconate (18472-51-0)
- Chlorobutanol (57-15-8) (C4H7Cl3O)
- Cyproheptadine Hydrochloride (969-33-5)
- Cytidylic Acid (63-37-6)
- Dicaffeine Malate (512.54 g/mol)
- Diclofenac Diethylamine (78213-16-8)
- Dextran (9004-54-0) (40000 g/mol)
- Diethylamine Salicylate (4419-92-5)
- Dextromethorphan (125-71-3) (C18H25NO)
- D-Galactose (59-23-4) (C6H12O6)
- Dibenzoyl-D-Tartaric Acid (17026-42-5)
- Dimethyl Fumarate (624-49-7)
- D-Xylose (58-86-6) (C5H10O5)
- Econazole Nitrate (24169-02-06)
- Enrofloxacin (93106-60-6) (C19H22FN3O3)
- Dimethylglycine (1118-68-9) (C4H9NO2)
- Diphenhydramine Hydrochloride (147-24-0)
- Erythritol (149-32-6) (C4H10O4)
- Doxycycline Hydrochloride (10592-13-9)
- Doxycycline Monohydrate (17086-28-1)
- Fructose (57-48-7) (C6H12O6)
- Gatifloxacin Sesquihydrate (180200-66-2)
- Gliclazide (21187-98-4) (C15H21N3O3S)
- Glimepiride (93479-97-1) (C24H34N4O5S)
- Glucosamine Hydrochloride (66-84-2)
- Glycine (56-40-6) (C2H5NO2)
- Ethylenediamine Dihydroiodide (5700-49-2)
- Famotidine (76824-35-6) (C8H15N7O2S3)
- Fenofibrate (49562-28-9) (C20H21ClO4)
- Ferric Ammonium Citrate
- Ferrous Ascorbate (24808-52-4)
- Ferrous Bisglycinate (20150-34-9)
- Inositol (87-89-8) (C6H12O6)
- Ferrous Gluconate (C12H22FeO14)
- L-Aspartic Acid (56-84-8)
- Flurbiprofen (5104-49-4) (C22H28Cl2F3N3OS)
- Folic Acid (59-30-3)
- Magnesium Gluconate (3632-91-5)
- Maize Starch (9005-25-8)
- Nimbin (5945-86-8) (C30H36O9)
- Adenosylcobalamin (13870-90-1) (C72H100CoN18O17P)
- Chlorogenic Acid (327-97-9)
- Colchicine (64-86-8) (C22H25NO6)
- Dasatinib (302962-49-8) (C22H26ClN7O2S)
- Dimenhydrinate (523-87-5) (C24H28ClN5O3)
- Dibenzoyl-L-Tartaric Acid (2743-38-6)
- Estradiol Anhydrous (50-28-2)
- Doxorubicin Hydrochloride (25316-40-9)
- Furosemide (54-31-9) (C12H11ClN2O5S)
- Gamma-Aminobutyric Acid (56-12-2)
- Glibenclamide (10238-21-8) (C23H28ClN3O5S)
- Ezetimibe (163222-33-1) (C24H21F2NO3)
- Polyvinylpyrrolidone (9003-39-8) ((C6H9NO)n)
- Methylcobalamin (13422-55-4) (C63H91CoN13O14P)
- Methylene Blue (61-73-4)
- Quinine Sulphate (C40H50N4O8S)
- Pectin (9000-69-5) (C6H10O7)
- Resveratrol (501-36-0) (C14H12O3)
- Risperidone (106266-06-2) (C23H27FN4O2)
- Saccharin (81-07-2) (C7H5NO3S)
- Selenious Acid (7783-00-8)
- Serine (56-45-1) (C3H7NO3)
- Sodium Stearyl Fumarate
- Tetraethylammonium Bromide (71-91-0)
- Thiamine Hydrochloride (67-03-8)
- Strontium Ranelate (C12H6N2O8SSr2)
- Strychnine Sulphate (60-41-3)
- Titanium Diboride (12045-63-5)
- Sulphamethoxazole (723-46-6) (C10H11N3O3S)
- Tranexamic Acid (1197-18-8)
- Taurine (107-35-7) (C2H7NO3S)
- Trientine (112-24-3) (C6H18N4)
- Valproic Acid (99-66-1)
- Vinpocetine (42971-09-05) (C22H26N2O2)
- Vitamin B12 (68-19-9)
- Vitamin D3 (67-97-0)
- Tripotassium Citrate (6100-05-06)
- Triprolidine Hydrochloride (6138-79-0)
- Atovaquone (95233-18-4) (C22H19ClO3)
- Curcumin (458-37-7) (C21H20O6)
- Fenbendazole (43210-67-9) (C15H13N3O2S)
- L-Lysine (56-87-1) (146.19 g/mol)
- N-Acetyl L-Cysteine (616-91-1)
- Octenidine Hydrochloride (70775-75-6)
- Piperidine (110-89-4) (C5H11N)
- Policosanol (142583-61-7) (494 g/mol)
- Pyridoxine Hydrochloride (58-56-0)
- Riboflavin (83-88-5) (C17H20N4O6)
- Terbutaline Sulfate (23031-32-5)
- Monosodium Citrate (18996-35-5)
- Di-Tert-Butyl Dicarbonate (24424-99-5)
- Quinine (130-95-0) (C20H24N2O2)
- Sodium Glycerophosphate (1334-74-3)
- Potassium Alginate (9005-36-1)
- Carrageenan (9000-07-01) (C23H23FN4O7Zn)
- Halquinol (8067-69-4) (C27H17Cl4N3O3)
- Magnesium L-Aspartate (C8H20MgN2O12)
- Povidone Iodine (25655-41-8)
- Sodium Polystyrene Sulfonate
- 2-1-Cyclohexenyl Ethylamine (3339-73-3)
- Sodium Saccharin (128-44-9)
- Phosphatidylserine (51446-62-9) (C42H82NO10P)
- Sodium Potassium Tartrate
- Di-P-Toluoyl-L-Tartaric Acid Monohydrate
- Thia mine
- High Fructose Corn Syrup
- Larch Turpentine Oil
- Magnesium Hydroxide Ip
- Econazole Nitrate Ip
- Sodium Iodide BP
- Calcium Lactate Gluconate
- Ferrous Fumarate (141-01-5)
- Thiopental Sodium (71-73-8)
- Policosanol Extract (Solid)
- Domperidone IP (C22H24ClN5O2)
- Vitamin D2 (50-14-6)
- Calcium Citrate Malate
- Vitamin A (68-26-8)
- L-Theanine (3081-61-6) (C7H14N2O3)
- L-Lysine Hydrochloride (657-27-2)
- 2-Acetylbutyrolactone (517-23-7) (C6H8O3)
- N-Hydroxy Succinimide (6066-82-6)
- Aluminum Acetate (C6H9AlO6)
- N,N-Diisopropylcarbodiimide (693-13-0) (C7H14N2)
- Acethydrazide (1068-57-1) (C2H6N2O)
- Tricalcium Phosphate IP
- Adenine (73-24-5) (C5H5N5)
- Corn Syrup (180.16 g/mol)
- 5-Amino-6-methylbenzimidazolone (67014-36-2) (C8H9N3O)
- Agar-Agar (9002-18-0) (C14H24O9)
- Dextromethorphan Hydrobromide (125-69-9)
- Betaine Anhydrous (107-43-7)
- Boric Acid IP
- Homoveratrylamine (120-20-7) (C10H15NO2)
- Bismuth Subgallate (99-26-3)
- Magnesium-L-Threonate (778571-57-6) (C8H14MgO10)
- Erdosteine (84611-23-4) (C8H11NO4S2)
- Trimethyl Chitosan (52349-26-5)
- Rabeprazole Sodium (117976-90-6)
- Nicotine Benzoate (88660-53-1)
- Mannitol (69-65-8) (C6H14O6)
- Linolenic Acid (463-40-1)
- Dextrose Monohydrate (14431-43-7)
- Ciprofloxacin Hydrochloride (93107-08-05)
- Uracil (66-22-8) (C4H4N2O2)
- Tyramine Hydrochloride (60-19-5)
- Zinc Monomethionine (56329-42-1)
- Zinc Gluconate (4468-02-04)
- Trospium Chloride (10405-02-04)
- Zinc Aspartate (36393-20-1)
- Tert-Butyl Bromoacetate (5292-43-3)
- Terbinafine Hydrochloride (78628-80-5)
- Tenofovir Disoproxil Fumarate
- Tadalafil (171596-29-5) (C22H19N3O4)
- Tizanidine Hydrochloride (64461-82-1)
- Tetracaine Hydrochloride (136-47-0)
- Sodium Butyrate (156-54-7)
- Sildenafil Citrate (171599-83-0)
- Sodium Alginate (9005-38-3)
- Salicylamide (65-45-2) (C7H7NO2)
- Simethicone (8050-81-5) (C6H18O4Si3)
- Quinine Hydrochloride (130-89-2)
- Potato Starch (9005-25-8)
- Diacerein (13739-02-01) (C19H12O8)
- Dehydrocholic Acid (81-23-2)
- Entecavir Anhydrous (142217-69-4)
- Epirubicin Hydrochloride (56390-09-01)
- Diosmin (520-27-4) (C28H32O15)
- Erythorbic Acid (89-65-6)
- Acesulfame Potassium (55589-62-3)
- Eszopiclone (138729-47-2) (C17H17ClN6O3)
- Ethacridine Lactate (1837-57-6)
- Dithranol (1143-38-0) (C14H10O3)
- Dopamine Hydrochloride (62-31-7)
- Fusidic Acid (6990-06-03)
- Gefitinib (184475-35-2) (C22H24ClFN4O3)
- Glycyrrhizin (1405-86-3) (C42H62O16)
- Fenticonazole Nitrate (73151-29-8)
- Ferric Carboxymaltose (9007-72-1)
- Finasteride (98319-26-7) (C23H36N2O2)
- Fluphenazine Hydrochloride (146-56-5)
- Polynoxylin (9011-05-06) ((C4H8N2O3)n)
- Pyrroloquinoline Quinone (72909-34-3)
- Racecadotril (81110-73-8) (C21H23NO4S)
- Reserpine (50-55-5) (C33H40N2O9)
- Ritonavir (155213-67-5) (C37H48N6O5S2)
- Rosmarinic Acid (20283-92-5)
- Roxithromycin (80214-83-1) (C41H76N2O15)
- Simvastatin (79902-63-9) (C25H38O5)
- Sodium Aminosalicylate (6018-19-5)
- Sertaconazole Nitrate (99592-39-9)
- Sevelamer Carbonate (845273-93-0)
- Tetracycline Hydrochloride (64-75-5)
- Sugammadex Sodium (343306-79-6)
- Tolvaptan (150683-30-0) (C26H25ClN2O3)
- Trandolapril (87679-37-6) (C24H34N2O5)
- Tamoxifen Citrate (54965-24-1)
- Tamoxifen Citrate
- Merbromin (129-16-8) (C20H8Br2HgNa2O6)
- Canola Oil (120962-03-0)
- Ascorbic Acid (50-81-7)
- Ammonium Acetate (631-61-8)
- Fish Oil (Liquid)
- Ta crolimus
- Paclitaxel IP (C47H51NO14)
- L- Thyroxine (51-48-9) (C15H11I4NO4)
- Zinc Citrate (546-46-3)
- Tannic Acid (1401-55-4)
- D-Pantothenic Acid Hemicalcium Salt
- G elatin
- Lignocaine Hydrochloride (73-78-9)
- 3,4-Diaminobenzoic acid (0619-05-06)
- Nicotine Salicylate (29790-52-1)
- Pentadecanoic Acid (1002-84-2)
- Oxytetracycline Hydrochloride (2058-46-0)
- Tri phosgene
- Tri methoprim
- Tr ibutyrin
- Suc ralose
- The obromine
- Stannous Pyrophosphate (15578-26-4)
- Sodium Ascorbate (644-95-6)
- Potassium Gluconate (299-27-4)
- Potassium Bitartrate (868-14-4)
- Free Flow Salt
- Ferrous Sulphate Anhydrous
- Guar Gum (9000-30-0)
- Glucono Delta Lactone
- Gluconic Acid (526-95-4)
- L- Arginine (74-79-3) (C6H14N4O2)
- I midazole
- Monosodium Glutamate Anhydrous
- Lactose Anhydrous (63-42-3)
- Lafutidine (118288-08-7) (C22H29N3O4S)
- Nicotine (54-11-5) (C10H14N2)
- L- Glutathione (70-18-8) (C10H17N3O6S)
- L- Proline (147-85-3) (C5H9NO2)
- Omeprazole (73590-58-6) (C17H19N3O3S)
- Paracetamol (103-90-2) (C8H9NO2)
- Potassium Iodate (2139718)
- Potassium Iodide (7681-11-0)
- Potassium Sorbate (24634-61-5)
- Miconazole Nitrate (22832-87-7)
- Microcrystalline Cellulose (9004-34-6)
- Rapeseed Oil (8002-13-9)
- 2,4-Dihydroxybenzoic Acid (89-86-1)
- Chloral Hydrate (302-17-0)
- Cyclohexylamine (108-91-8) (C6H13N)
- Cysteamine Hydrochloride (156-57-0)
- Diclofenac Sodium (15307-79-6)
- Degarelix Acetate (934016-19-0)
- Dextrin (9004-53-9) (C18H32O16)
- Dimethyl Sulfoxide (67-68-5)
- Disulfiram (97-77-8) (C10H20N2S4)
- D-Mandelic Acid (611-71-2)
- D-Tartaric Acid (147-71-7)
- Imidazole (288-32-4) (C3H4N2)
- Triphosgene (32315-10-9) (C3Cl6O3)
- Trimethoprim (738-70-5) (C14H18N4O3)
- Tributyrin (60-01-5) (C15H26O6)
- Sucralose (56038-13-2) (C12H19Cl3O8)
- Theobromine (83-67-0) (C7H8N4O2)
- Thiamine (C12H17N4OS+) (Thiamin)
- Tacrolimus (104987-11-3) (C44H69NO12)
- Gelatin (9000-70-8) (100000 g/mol)
- Isomalt (64519-82-0) (C12H24O11)
- Starch (100000 g/mol) (300 C)
- 2-Chloro-7,8-Dihydroquinolin-5(6H)-One (124467-36-3) (C9H8ClNO)
- Brimonidine Tartrate (70359-46-5)
- 5-Bromo-1H-Indazole (53857-57-1) (C7H5BrN2)
- 5-Sulphosalicylic Acid Dihydrate
- Miglustat Hydrochloride (210110-90-0)
- Mometasone Furoate (83919-23-7)
- Rac-Nicotine (22083-74-5) (C10H14N2)
- Deoxycholic Acid (83-44-3)
- Desflurane (57041-67-5) (C3H2F6O)
- Ferric Pyrophosphate Anhydrous
- Salbutamol (18559-94-9) (C13H21NO3)
- Calcium Iodide (10102-68-8)
- lSerine (56-45-1) (C3H7NO3)
- 4-Bromoacetophenone (99-90-1) (C8H7BrO)
- Acetic Acid Food Grade
- Aminoguanidine Bicarbonate (2582-30-1)
- Ascorbic Acid BP
- Ascorbic Acid IP
- Benzyl Benzoate IP
- Caffeine Sodium Benzoate
- Calcium Ascorbate USP
- Calcium Chloride Food Grade
- Calcium Citrate USP
- Canola Oil Food Grade
- Chlorphenesin IP (184.62 g/mol)
- Conjugated Linoleic Acid Powder
- Dichlorophene (97-23-4) (C13H10Cl2O2)
- Diiodohydroxyquinoline (83-73-8) (C9H5I2NO)
- Ethyl Oleate Ip
- Heparin Sodium IP
- Hydrobromic acid 48% Solution
- Hydroxypropyl Methyl Cellulose 15 CPS
- L-Tyrosine (60-18-4) (C9H11NO3)
- Levothyroxine Sodium (55-03-8)
- Magnesium Glycinate Food Grade
- Microcrystalline Cellulose 101
- Microcrystalline Cellulose 102
- N-Octyl Gallate (1034-01-1)
- Nandrolone Decanoate (360-70-3)
- Potassium Gluconate Food Grade
- Selenium Yeast (196.25 g/mol)
- Sodium Ascorbate IP
- Sodium Ascorbate Ip Mesh100
- Sodium citrate Food Grade
- Sodium Selenate Food Grade
- Sodium Starch Glycolate BP
- Sodium Thiomethoxide (CH3NaS)
- Tetrasodium Pyrophosphate Food Grade
- Trehalose (99-20-7) (C12H22O11)
- Tripotassium EDTA (17572-97-3)
- Vitamin K2 (863-61-6)
- Yohimbine Hydrochloride (65-19-0)
- PEG - 300 (Polyethylene Glycol 300)
- PEG - 400 (Polyethylene Glycol 400)
- PEG - 600 (Polyethylene Glycol 600)
- PEG - 1000 (Polyethylene Glycol 1000)
- PEG - 2000 (Polyethylene Glycol 2000)
- PEG - 4000 (Polyethylene Glycol 4000)
- PEG - 6000 (Polyethylene Glycol 6000)
- Palmitic Acid 85%
- N,N-Dimethylpropyleneurea (7226-23-5) (C5H12N2O)
- Monomethylamine 25% In Meoh
- Monomethylamine 30% In Meoh
- Tri-N-Butylamine (102-82-9) (Tributylamine)
- Cis-Tosylate (154003-23-3) (Solid)
- Ethyl 4-Bromobutyrate (2969-81-5)
- Ethyl 4-Dimethylaminobutyrate (22041-23-2)
- 2-Amino-3,5-Dibromobenzaldehyde (50910-55-9) (3,5-Dibromo-2-Aminobenzaldehyde)
- Bromo OTBN (114772-54-2)
- R-Mandelic Acid (611-71-2)
- 1,7-Dibromoheptane (4549-31-9) (C7H14Br2)
- Ethyl-2,2-Dibromopropionate (34757-17-0) (C5H8Br2O2)
- 11-Bromoundecanoic Acid (01-05-2834)
- 1-Bromodecane (112-29-8) (C10H21Br)
- 1-Bromo-2,4-Difluorobenzene (348-57-2) (C6H3BrF2)
- Ethyl-2-Bromobutyrate (533-68-6) (C6H11BrO2)
- 1-Bromo-2,5-Difluorobenzene (399-94-0) (C6H3BrF2)
- Ethyl-3-Bromopropionate (539-74-2) (4-Bromobutyric Acid Ethyl Ester)
- 1-Bromo 3,4-Difluorobenzene (348-61-8) (3,4-Difluorobromobenzene)
- 1-Bromo-3,5-Dichlorobenzene (19752-55-7) (C6H3BrCl2)
- Aroma Chemicals
- Home Page
- Company Profile
- Contact Us

- Home
- Products
- Industrial Chemicals
- Sodium Amide (7782-92-5)
Sodium Amide (7782-92-5)
Price 325.0 INR/ Kilograms
Sodium Amide (7782-92-5) Specification
- Appearance
- White crystalline solid
- Molecular Weight
- 39.01 g/mol Grams (g)
- Structural Formula
- NaNH2
- CAS No
- 7782-92-5
- Refractive Rate
- 1.5
- Physical Form
- Solid
- Purity
- 98 99%
- Properties
- Sodium amide is a powerful, highly reactive inorganic base primarily used in organic synthesis. It is a critical reagent for dehydrohalogenation reactions and the synthesis of alkynes. Due to its extreme sensitivity to moisture and air, it must be handled under inert atmospheric conditions in industrial laboratories.
- Usage
- Strong base used in organic synthesis for deprotonation of terminal alkynes to generate acetylide anions; used to prepare amines; moisture-sensitive; stored under inert atmosphere.
- Solubility
- Insoluble in water; reacts with water to form ammonia and sodium hydroxide.
- Molecular Formula
- NaNH2
- Melting Point
- 210 C
- Application
- Industrial
Sodium Amide (7782-92-5) Trade Information
- Minimum Order Quantity
- 25 Kilograms
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Kilograms Per Month
- Delivery Time
- 10 Days
- Main Domestic Market
- All India
About Sodium Amide (7782-92-5)
Powerful Base for Organic Synthesis
Sodium amide is widely recognized as one of the strongest inorganic bases used in industrial and laboratory organic synthesis. Its capacity to deprotonate terminal alkynes and initiate various critical transformations makes it invaluable for the production of alkynes, amines, and related compounds. Its efficiency and reliability in facilitating extensive organic reactions are key assets in chemical production.
Handling and Storage Precautions
Given its extreme reactivity, sodium amide must be handled with rigorous care. Exposure to moisture or air can cause hazardous reactions, leading to the generation of ammonia and sodium hydroxide. It is essential to store sodium amide under an inert atmosphere, such as nitrogen or argon, in tightly sealed containers, away from any sources of water or humidity to preserve its purity and safety.
FAQ's of Sodium Amide (7782-92-5):
Q: How should sodium amide be handled and stored safely?
A: Sodium amide must be handled under an inert atmosphere, such as nitrogen or argon, due to its sensitivity to air and moisture. It should be stored in tightly sealed containers in a dry, cool area to prevent contact with water or humidity, which could trigger hazardous reactions.Q: What are the primary industrial uses of sodium amide?
A: Sodium amide is primarily utilized as a strong base in organic synthesis, especially for deprotonating terminal alkynes and generating acetylide anions. It also facilitates dehydrohalogenation reactions and the synthesis of amines, making it an essential reagent in chemical manufacturing.Q: When is sodium amide chosen over other bases in organic reactions?
A: Sodium amide is selected when a particularly strong base is required, such as in the deprotonation of terminal alkynes or when generating nucleophilic species like acetylide anions, due to its high reactivity and effectiveness in facilitating transformations that weaker bases cannot achieve.Q: Where is sodium amide commonly used?
A: Sodium amide finds widespread use in industrial chemical laboratories and manufacturing facilities across India and globally, particularly in the synthesis of fine chemicals, pharmaceuticals, and various organic compounds.Q: What process occurs when sodium amide reacts with water?
A: Upon contact with water, sodium amide undergoes a vigorous reaction, producing ammonia gas and sodium hydroxide, both of which are hazardous. Therefore, it is considered insoluble in water due to this rapid chemical reaction rather than simple dissolution.Q: What benefits does sodium amide offer in organic synthesis?
A: Its major benefit is its strength and reliability as a base, enabling unique synthon formation and facilitating the efficient synthesis of complex molecules, which are otherwise challenging with less reactive bases.

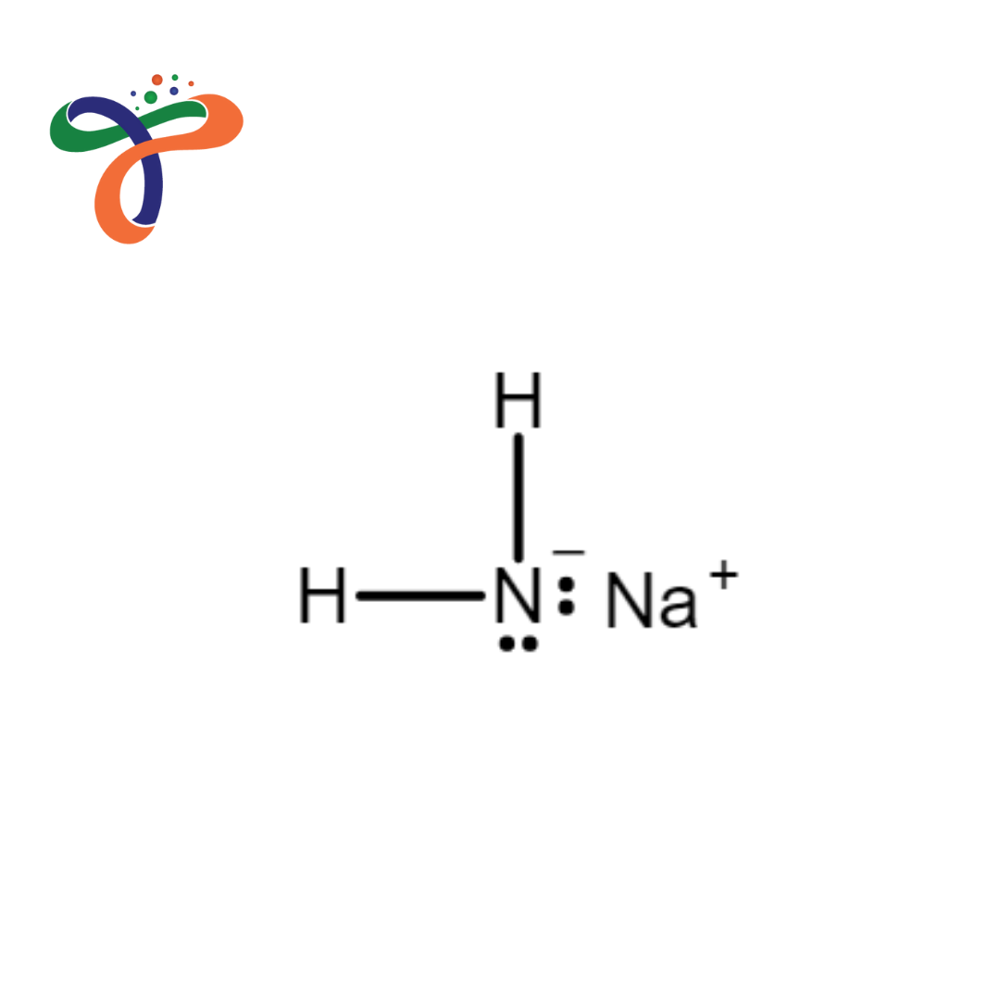

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+

More Products in Industrial Chemicals Category
Monoisopropanolamine (78-96-6) (C3H9NO)
Price 210.0 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Appearance : Colorless to pale yellow liquid
Molecular Weight : 75.11 g/mol Grams (g)
Structural Formula : C3H9NO
CAS No : 78966
Tungstic Acid (7783-03-01)
Price 8750.0 INR
Minimum Order Quantity : 25
Appearance : Yellow powder
Molecular Weight : 249.86 g/mol Grams (g)
Structural Formula : H2O4W
CAS No : 7783031
3-Hydroxy-N-Phenyl-2-Naphthamide (92-77-3) (C17H13NO2)
Price 360.0 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Appearance : Yellowbrown crystalline solid
Molecular Weight : 263.30 g/mol Grams (g)
Structural Formula : C17H13NO2
CAS No : 92773
Graphite (7782-42-5) (C)
Price 55.0 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Appearance : Dark gray to black with metallic luster
Molecular Weight : 12.01 g/mol Grams (g)
Structural Formula : C
CAS No : 7782425



Thank You!
Thank You for your valuable time. We have received your details and will get back to you shortly.
Our Products

Developed and Managed by Infocom Network Private Limited.

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese